Private: Main Body
11 Archaic Homo
Amanda Wolcott Paskey, M.A., Cosumnes River College
AnnMarie Beasley Cisneros, M.A., American River College
Learning Objectives
- Describe the unique anatomical and cultural characteristics of archaic Homo sapiens in contrast to other hominins.
- Articulate how archaic Homo sapiens fossils fit into anatomical evolutionary trends including brain size development, as well as cultural innovations and distribution throughout the Old World.
- Explain how shifting environmental conditions required flexibility of adaptations, both anatomically and culturally, for hominin survival and the potential consequences of a high degree of specialization.
- Recognize that while archaic Homo sapiens share similarities, they are characterized by significant regional variation and local adaptation.
- Detail the increased complexity and debates surrounding archaic Homo sapiens’ classification in light of transitional species, species admixture, etc.
BREAKING THE STIGMA OF THE “CAVEMAN”

What do you think of when you hear the word “caveman”? Perhaps you imagine a character from a film such as The Croods, Tarzan, or Encino Man or from the cartoon The Flintstones. Maybe you picture the tennis-playing, therapy-going hairy Neanderthals from Geico Insurance commercials. Or perhaps you imagine comic characters from “The Far Side” or “B.C.” comics. Whichever you picture, the character in your mind is likely stooped over with a heavy brow, tangled long locks and other body hair, and clothed in animal skins, if anything. They might be holding a club with a confused look on their face, standing at the entrance to a cave or dragging an animal carcass to a fire for their next meal (see Figure 11.1). You might have even signed up to take this course because of what you knew—or expected to learn—about “cavemen.”
These images have long been the stigma and expectation about our ancestors at the transition to modern Homo sapiens. Tracing back to works as early as Linnaeus, scientists once propagated and advanced this imagery, creating a clear picture in the minds of early scholars that informed the general public, even through today, that archaic Homo sapiens, “cavemen,” were somehow distinctly different and much less intelligent than we are now. Unfortunately, this view is incorrect, overly simplistic, and misleading. Understanding what archaic Homo sapiens were actually like requires a much more complex and nuanced picture, one that continues to be understood with greater clarity as current research uncovers the lives of our not-too-distant (and not-too-different) ancestors.
The first characterizations of archaic Homo sapiens were formed from limited fossil evidence in a time when and species-centric perspectives () were more extensively accepted and entrenched in both society and science. Today, scientists are working from a more complete fossil record from three continents (Africa, Asia, and Europe) and even genetic evidence that informs their analyses and conclusions. The existence of archaic Homo sapiens mark an exciting point in our lineage—a point at which many modern traits had emerged and key refinements were on the horizon. Anatomically, we today are not that much different from archaic Homo sapiens.
This chapter will examine the environment with which archaic Homo sapiens had to contend, one that shaped their, and our, ultimate development. It will also examine the key anatomical traits that define this group of fossils (including the infamous subgroup known as Neanderthals), cultural innovations that aided their adaptation to the changing environment, and their geographic distribution and regional variations. Additionally, it will consider areas of exciting new research that suggest even greater nuance and complexity during this time period. Our understanding of this species and our evolution is complex and exciting and could become even more so as new data is uncovered.
THE CHANGING ENVIRONMENT
While modern climate change is of critical concern today due to its cause (human activity) and pace (unprecedentedly rapid), the existence of global climate change itself is not a recent phenomenon. The climate across the globe has changed, drastically at times, over the course of Earth’s existence. The Pleistocene epoch—between 1.8 million years ago (mya) and 11,000 years ago (kya); illustrated in Figure 7.7—was a time of great climatic upheaval. The Middle Pleistocene, roughly between 780 kya and 125 kya, is the time period in which archaic Homo sapiens appear in the fossil record—a time that witnessed some of the most drastic climatic changes that have been seen in human existence. During this time period, there were 15 major and 50 minor glacial events in Europe alone!
What exactly is a ? When scientists talk about glacial events, they are referring to the climate being in an ice age. This means that the ocean levels were much lower than today, as much of the earth’s water was tied up in large glaciers or ice sheets. Additionally, the average temperature would have been much cooler, which would have better supported an Arctic or tundra-adapted plant-and-animal ecosystem in northern latitudes. The most interesting and relevant features of Middle Pleistocene glacial events are the sheer number of them and their repeated bouts—this era alternated between glacial periods and warmer periods, known as . In other words, the world wasn’t in an ice age the whole time.
How have scientists determined how many glaciations there were during the Middle Pleistocene and how severe they were? Several lines of evidence help inform our understanding of past climates. One important source is the study of oxygen isotopes preserved in the shells of marine invertebrates called . Foraminifera incorporate oxygen from seawater during their life. When they die, foraminifera shells fall to the ocean floor and can be preserved as microscopic fossils that are part of the sediment, which can later be sampled and studied in . Studying these cores has revealed that the oxygen isotope present varies depending on Earth temperatures at the time the foraminifera were alive. During glacial periods, seawater is cooler and one oxygen isotope—18O—is in higher concentrations in seawater (and, as a result, in foraminifera shells) because it is heavier. In contrast, water with the oxygen isotope 16O is lighter and therefore evaporates first, becomes part of precipitate (such as snow), and eventually becomes trapped in glaciers. During interglacials, 16O returns to the ocean in water runoff, resulting in higher ocean and foraminifera concentrations of this oxygen isotope. Recent research with further confirms the length and severity of glacial periods using similar techniques.
The Pleistocene is characterized by shifts in Earth’s temperatures and their impact on plant and animal life. The Middle Pleistocene was an even more intense period of fluctuation with frequent and severe glacial and interglacial episodes recorded in marine isotopes, among other data points. You can see the dramatic and increasing fluctuations in temperature, recorded through foraminifera, in the chart (Figure 11.2). The distance between lows and highs demonstrates the severity of temperature shift. Much as the Richter scale represents more intense earthquakes with more dramatic peaks, so too does this chart, which uses dramatic peaks to demonstrate intense temperature swings.

Glacial periods are defined by Earth’s average temperature being lower. Worldwide, temperatures are reduced, with cold areas becoming even colder. The water cycle experiences limited runoff as water evaporates from the seas, precipitates (often as snow and ice), and accumulates in glaciers with little precipitation melting as runoff. Over time, continued evaporation with little runoff results in the accumulation of snowpacks and glaciers at the expense of sea level, which is lowered. In simple terms, the water that is normally in the sea is now tied up on land as ice. Huge portions of the landscape may have become inaccessible during glacial events due to the formation of glaciers and massive ice sheets. In Europe, the Scandinavian continental glacier covered what is today Ireland, England, Sweden, Norway, Denmark, and some of continental Europe. Plant and animal communities shifted to lower latitudes along the periphery of ice sheets. Additionally, some new land was opened during glacials. Evaporation with little runoff reduced sea levels by as much as almost 150 meters, shifting coastlines outward by in some instances as much as almost 100 kilometers. Additionally, land became exposed that connected what were previously unconnected continents such as Africa at the Gulf of Aden into Yemen.
Glacial periods also affected equatorial regions and other regions that are today thought of as warmer or at least more temperate parts of the globe, including Africa. While these areas were not covered with glaciers, the impact of increased global glaciation resulted in lower sea levels and expanded coastlines. Cooler temperatures were accompanied by the drying of the climate, which caused significantly reduced rainfall, increased aridity, and the expansion of deserts. It is an interesting question to consider whether the same plants and animals that lived in these regions prior to the ice ages would be able to survive and thrive in this new climate? Plant and animal communities shifted in response to the changing climate, whenever possible.
Rather than a single selective force, the Middle Pleistocene was marked by periods of fluctuation, not just cold periods. Interglacials interrupted glaciations, reversing trends in sea level, coastline, temperature, precipitation, and aridity, as well as glacier size and location. Interglacials are marked by increased rainfall and a higher temperature, which causes built-up ice in glaciers to melt. Interglacials are marked by glacial retreat, which is the shrinking of glaciers and the movement of the glaciers back toward the poles, as we’ve seen in our lifetime. During interglacials, sea levels increase, flooding some previously exposed coastlines and continental connections. In addition, plant and animal communities shift accordingly, often finding more temperate climates to the north and less arid and more humid climates in the tropics.
Scientists have found that at one site, the Olorgesailie region in southern Kenya, a single location was at various times in the Middle Pleistocene a deep lake, a drought-dried lakebed, small streams, and a grassland. While various animal species would have moved in and out of the area as the climate shifted, some animal species went extinct, and new, often related, species took up residence. The trend, scientists noted, was that animals with more specialized features went extinct and animals with more generalized features, such as animals we see today, survived in this changing climatic time period. For example, a zebra with specialized teeth for eating grass was ultimately replaced by a zebra that could eat grass and other types of vegetation. The exclusively terrestrial fossil baboon Therapithecus oswaldi was replaced by Papio anubis, the more flexible locomotor baboon that exists in the region today. If this small, localized example shows such a dramatic change in terms of the environment and the plant and animal biocommunities, what would have been the impact on humans?
There is no way humans could have escaped the effects of Middle Pleistocene climate change, no matter what region of the world they were living in. As noted earlier, and as evidenced by what was seen in the other biotic communities, humans would have faced changing food sources as previous sources of food may have gone extinct or moved to a different latitude. Depending on where they were living, fresh water may have been limited. Durial glacials, lower sea levels would have given humans more land to live on, while the interglacials would have reduced the available land through the increase in rainfall and associated sea level rise. Dry land connections between the continents would have made movement from one continent to another by foot easier at times than today, although these passageways were not consistently available through the Middle Pleistocene due to the glacial/interglacial cycle. Finally, as evidenced by the study at the Olorgesailie region in Kenya, during the Middle Pleistocene animal species that were overly specialized to one particular type of environment were less likely to survive when compared to their more generalized counterparts. Evidence suggests that this same pattern may have held true for archaic Homo sapiens, in terms of their ability to survive this dramatic period of climate change.
DEFINING CHARACTERISTICS OF ARCHAIC HOMO SAPIENS
Archaic Homo sapiens share our species name but are distinguished by the term “archaic” as a way of recognizing both the long period of time between their appearance and ours, as well as the way in which human traits have continued to evolve over time—making archaic Homo sapiens look slightly different from us today, despite technically being considered the same species. Living throughout the Old World during the Middle Pleistocene, archaic Homo sapiens are considered, in many ways, transitional between Homo erectus and modern Homo sapiens (see Figure 11.3). All archaic Homo sapiens share the defining trait of an increased brain size—specifically a brain of at least 1,100 cc and averaging 1,200 cc—but are also characterized by significant regional and temporal (time) variations. Because of these variations, scientists disagree on whether these fossils represent a single, variable species or multiple, closely related species (sometimes called Homo antecessor, Homo heidelbergensis, Homo georgicus, Homo neanderthalensis, and Homo rhodesiensis). For simplicity we are going to lump them all together under the heading of archaic Homo sapiens and discuss them as a unit, with the exception of a particularly unique and well-known population living in Europe and West Asia known as the Neanderthals, which we will examine separately.
|
Trait |
Homo erectus |
Archaic Homo sapiens |
Anatomically Modern Homo sapiens |
|
Average Brain Size |
900 cc |
1,200 cc (1,500 cc when including Neanderthals) |
1,400 cc |
|
Skull Shape |
Long and low Angular |
Intermediate |
Short and high
|
|
Forehead |
Absent |
Emerging |
Present |
|
Nasal Region |
Projecting nasal bones (bridge of the nose), no midfacial prognathism |
Wider nasal aperture and midfacial prognathism |
Narrower nasal aperture, no midfacial prognathism |
|
Chin |
Absent |
Absent |
Present |
|
Other Facial Features |
Large brow ridge and large projecting face |
Intermediate |
Small brow ridge and |
|
Other Skull Features |
Nuchal torus, sagittal keel, thick cranial bone |
Projecting occipital bone, often called occipital bun in Neanderthals; intermediate thickness of cranial bone |
Small bump on rear of skull, if anything; thin cranial bone |
|
Dentition |
Large teeth, especially front teeth |
Slightly smaller teeth; front teeth still large; retromolar gap in Neanderthals |
Smaller teeth |
|
Postcranial Features |
Robust bones of skeleton |
Robust bones of skeleton |
More gracile bones of skeleton |
Figure 11.3 A comparison of Homo erectus, archaic Homo sapiens, and anatomically modern Homo sapiens. This table compares key traits of the crania and postcrania that distinguish these three hominins.
When comparing Homo erectus, archaic Homo sapiens, and anatomically modern Homo sapiens across several anatomical features, one can see quite clearly that archaic Homo sapiens are intermediate in their physical form. This follows the trends first seen in Homo erectus for some features and in other features having early, less developed forms of traits more clearly seen in modern Homo sapiens. For example, archaic Homo sapiens trended toward less angular and higher skulls than Homo erectus but had skulls notably not as short and globular in shape and with a less developed forehead than anatomically modern Homo sapiens. archaic Homo sapiens had smaller brow ridges and a less-projecting face than Homo erectus and slightly smaller teeth, although incisors and canines were often about as large as that of Homo erectus. Archaic Homo sapiens also had a wider , or opening for the nose, as well as a forward-projecting midfacial region, known as . The occipital bone often projected and the cranial bone was of intermediate thickness, somewhat reduced from Homo erectus but not nearly as thin as that of anatomically modern Homo sapiens. The postcrania remained fairly robust, as well. To identify a set of features that is unique to the group archaic Homo sapiens is a challenging task, due to both individual variation—these developments were not all present to the same degree in all individuals—and the transitional nature of their features. Neanderthals will be the exception, as they have several clearly unique traits that make them notably different from modern Homo sapiens as well as their closely related archaic cousins.

The one thing that is clear about archaic Homo sapiens is that regional variation, first seen in the different Homo erectus specimens across Asia and Africa, is clearly present and even more pronounced. While the general features of archaic Homo sapiens, identified earlier, are present in the fossils of this time period, there are significant regional differences. The majority of this regional variation lies in the degree to which fossils have features more closely aligned with Homo erectus or with anatomically modern Homo sapiens.

To illustrate this point, we will examine three exemplary specimens, one from each of the three continents on which archaic Homo sapiens lived. In Africa, “Broken Hill Man,” one of several individuals found in the Kabwe lead mine in Zambia, had a large brain (1,300 cc) and taller cranium as well as many Homo erectus-like skull features, including massive brow ridges, a large face, and thick cranial bones (Figure 11.4). Conditions for preservation in Asia during the Middle Pleistocene were not as conducive to the fossilization of complete crania; however, many archaic Homo sapiens skullcaps have been found. One partial crania from Dali, China, is representative of archaic Homo sapiens in Asia, including large and robust features with heavy brow ridges, akin to what is seen in Homo erectus, and a large cranial capacity intermediate between Homo erectus and anatomically modern Homo sapiens. Across Europe, many near-complete archaic Homo sapiens crania have been discovered, including one, part of an almost-complete skeleton, found in northern Spain at Atapuerca. Atapuerca 5 (Figure 11.5) has thick cranial bone, an enlarged cranial capacity, intermediate cranial height, and a more rounded cranium than seen previously. Additionally, Atapuerca 5 demonstrates features that foreshadow Neanderthals, including increased midfacial prognathism. After examining some of the fossils, the transitional nature of archaic Homo sapiens is clear—their features place them squarely between Homo erectus and modern Homo sapiens.
Due to the transitional nature of archaic Homo sapiens, identifying the time period they are associated with is problematic and complex. Generally, it is agreed upon that archaic Homo sapiens lived between 600,000 and 200,000 years ago. But regionally this varies with considerable overlap between Homo erectus on the early end of the spectrum and modern Homo sapiens and Neanderthals on the latter end. The earliest-known archaic Homo sapiens fossils tentatively date to about 600,000 years ago in Africa, whereas archaic Homo sapiens fossils in Asia appear around 300,000 years ago and in Europe around 350,000 years ago (and potentially as early as 600,000 years ago). The end point of archaic Homo sapiens is also problematic since it largely depends upon when the next subspecies of Homo sapiens appears and the classification of highly intermediate specimens. For example, in Africa, the end of archaic Homo sapiens is met with the appearance of modern Homo sapiens, while in Europe it is the appearance of Neanderthals that is traditionally seen as the end of archaic Homo sapiens.
Archaic Homo sapiens mark an important chapter in the human lineage, bridging more ancestral forms, such as Homo erectus, with modern Homo sapiens. During this period of climatic transition and fluctuation, archaic Homo sapiens mirror the challenges of their environments. Showing increasing regional variation due to the need for local adaptation, there is no single archetype for this group but, rather, multiple variations; their transitional nature is one of their key defining characteristics.
NEANDERTHALS
One particularly well-known population of archaic Homo sapiens are the Neanderthals, named after the site where they were first discovered in the Neander Valley, or “thal” in German, located near Dusseldorf, Germany. Popularly known as the stereotypical “cavemen” examined at the outset of this chapter, recent research is upending long-held beliefs about this group of archaics. As mentioned in the introduction, Neanderthals have long been thought to be dumb brutes who were, behaviorally speaking, not much different from apes. However, today, scientists agree that Neanderthal behavior was increasingly complex and nuanced, far beyond what was exhibited by even other archaic Homo sapiens discussed throughout this chapter. We implore you to forget the image of the iconic caveman and have an open mind when exploring the fossil evidence of the Neanderthals.
It is important to understand why Neanderthals are typically separated from other archaic Homo sapiens. Unlike the rest of archaic Homo sapiens, Neanderthals are easily defined and identified in many ways. There is a clear geographic boundary of where Neanderthals lived: western Europe, the Middle East, and western Asia. The time period for when Neanderthals lived is widely accepted as between 150,000 and 35,000 years ago. Additionally, Neanderthals have a unique and distinct cluster of physical characteristics. While a few aspects of Neanderthals are less clear cut and are shared among some archaic Homo sapiens, such as the types of tools they created and used, most attributes of Neanderthals, both anatomically and behaviorally, are unique to them.
As mentioned previously, the geographic distribution of Neanderthals is very specific. Neanderthal fossils, thus far, have been found across a narrow latitude of western Europe, the Middle East, and western Asia. No Neanderthal fossils have ever been discovered outside of this area, including Africa. This is a bit curious, as other archaics seem to have adapted in Africa and then migrated elsewhere, but Neanderthals’ regional association makes sense in light of the environment to which they were best adapted. While Neanderthals lived in different ecosystems, including temperate environments, they were very well adapted to extreme cold weather and their geographic distribution includes what would have been some of the coldest habitable locations at the time of their existence.
Neanderthals lived during some of the coldest times during the last Ice Age and at far northern latitudes. This means Neanderthals were living very close to the glacial edge, and not in a more temperate region of the globe, like some of their archaic Homo sapiens relatives. Their range likely expanded and contracted along with European glacial events, moving into the Middle East during glacial events when Europe became even cooler, and when the animals they hunted would have moved for the same reason. During interglacials, when Europe warmed a bit, Neanderthals and their prey would have been able to move back into Western Europe.
Many of the Neanderthals’ defining physical features are more extreme and robust versions of traits seen in other archaic Homo sapiens, clustered in this single population (Figure 11.6). Brain size is one of the Neanderthal features that continues to follow the same patterns as seen with other archaic Homo sapiens, namely an enlargement of the cranial capacity. The average Neanderthal brain size is around 1,500 cc, and the range for Neanderthal brains can extend to upwards of 1,700 cc. The majority of the increase in the brain occurs in the occipital region, or the back part of the brain, resulting in a skull that has a large cranial capacity with a distinctly long and low shape that is slightly wider than previous forms at far back of the skull. Modern humans have a brain size comparable to that of Neanderthals; however, our brain expansion occurred in the frontal region of the brain, not the back, as in Neanderthal brains. This difference is also the main reason why Neanderthals lack the vertical forehead that modern humans possess. They simply did not need an enlarged forehead, because their brain expansion occurred in the rear of their brain. Due to cranial expansion, the back of the Neanderthal skull is less angular (as compared to Homo erectus) and is more rounded, a feature similar to that of modern Homo sapiens.
Another feature that continues the trend noted in previous hominins is the enlargement of the nasal region, or the nose. Neanderthal noses are large and have a wide nasal aperture, which is the opening for the nose. While the nose is only made up of two bones, the nasals, the true size of the nose can be determined by looking at other facial features, including the nasal aperture, and the angle of the nasal and maxillary, or facial bones. In Neanderthals, these indicate a large, forward-projecting nose that appears to be pulled forward away from the rest of the face. This feature is further emphasized by the backward-sloping nature of the cheekbones, or the zygomatic arches. The unique shape and size of the Neanderthal nose is often characterized by the term midfacial prognathism—a jutting out of the middle portion of the face, or nose. This is in sharp contrast to the prognathism exhibited by other hominins, who exhibited prognathism, or the jutting out, of their jaws.
The teeth of the Neanderthals follow a similar pattern seen in the archaic Homo sapiens, which is an overall reduction in size, especially as compared to the extremely large teeth seen in the genus Australopithecus. However, while the teeth have continued to reduce, the jaw size does not keep pace, leaving Neanderthals with an interesting situation. Their jaw is oversized for their teeth, leaving a gap between their final molar and the end of their jaw. This gap is called a .
The projecting occipital bone present in other archaic Homo sapiens is also more prominent in Neanderthals, extending the trend found in archaics. Among Neanderthals, this projection of bone is easily identified by its bun shape on the back of the skull and is known as an . This projection appears quite similar to a dinner roll in size and shape. Its purpose, if any, remains unknown.
Continuing the archaic Homo sapiens trend, Neanderthal brow ridges are prominent but somewhat smaller in size than those of Homo erectus and earlier archaic Homo sapiens. In Neanderthals, the brow ridges are also often slightly less arched than those of other archaic Homo sapiens.
In addition to extending traits present in archaic Homo sapiens, Neanderthals possess several distinct traits. Neanderthal , the holes in the maxillae or cheek bones through which blood vessels pass, are notably enlarged compared to other hominins. The Neanderthal postcrania are also unique in that they demonstrate increased robusticity in terms of the thickness of bones and body proportions that show a barrel-shaped chest and short, stocky limbs, as well as increased musculature. These body portions are seen across the spectrum of Neanderthals—in men, women, and children.
Many of the unique traits that Neanderthals possess can be attributed to adaptation to the extreme cold environments in which they often lived. Together explained as cold adaptations, these traits are thought to be a response to the cold, dry environments in which Neanderthals lived and which certainly exerted strong selective forces. For example, Bergmann’s and Allen’s Rules dictate that an increased body mass and short, stocky limbs are common in animals that live in cold conditions. Neanderthals match the predictions of Bergmann’s and Allen’s Rules perfectly. In addition, the Neanderthal skull also exhibits adaptations to the cold. Neanderthals’ large infraorbital foramina allow for larger blood vessels, increasing the volume of blood that is found closest to the skin, which helps to keep the skin warmer. The midfacial prognathism present in Neanderthals indicates that Neanderthals would have had a large nose. This enlarged nose may also have been beneficial to have in cold weather due to longer nasal passages and mucus membranes for cold air to travel through before reaching the lungs. It is very uncomfortable and challenging to breathe and exert oneself in exceptionally cold, dry air. The more time the air spends in the mucus membranes, the warmer and more moist the air will be before it reaches the lungs. The Neanderthals’ larger nose has long been thought to have acted as a humidifier, easing physical exertion in their climate, although research on this particular trait continues to be studied and debated.
|
Distinct Neanderthal Anatomical Features |
|
|
Brain Size |
1,500 cc average |
|
Skull Shape |
Long and low |
|
Brow Ridge Size |
Large |
|
Nose Size |
Large, with midfacial prognathism |
|
Dentition |
Reduced, but large jaw size, creating retromolar gap |
|
Occipital Region |
Enlarged occipital region, occipital bun |
|
Other Unique Cranial Features |
Large infraorbital foramina |
|
Postcranial Features |
Short and stocky body, increased musculature, barrel-shaped chest |
Figure 11.6 Neanderthal distinguishing features. This table outlines key features associated with Neanderthals.

In summary, Neanderthal characteristics are a distinct cluster of features, some of which were apparent in previous hominins and others that were unique. Additionally, it is clear that Neanderthals were specially adapted to a particular environment—a very cold one. A classic example of a Neanderthal with all of the characteristics mentioned above is the La Ferrassie 1 Neanderthal, from France. The skeleton is near complete, which is not necessarily unique among Neanderthal fossils as many partially complete remains have been found, but it does provide us with a lot of information. The La Ferrassie 1 Neanderthal, who was male, had a brain size of around 1640cc and had an extremely large nose and infraorbital foramina. Additionally, the brow ridges are marked in size, and the overall skeleton is robust (Figure 11.7).
What are the benefits or the potential challenges Neanderthals could have faced for being highly specialized to one particular environment, when we know their environment and climate were in flux?
Neanderthal Culture and Lifeways
One key Neanderthal adaptation was their cultural innovations. Cultural innovation is a key way that hominins adapt to their environment. As you recall, the culture of Homo erectus was marked by the development of a bifacial tool, the Acheulean handaxe, which allowed them differential access to meat on animal carcasses when compared to their predecessors. For Homo erectus, the Acheulean handaxe allowed more efficient removal of meat and possibly calculated scavenging. The increase in their body and brain size, along with their more effective tools, allowed them to track predators and snatch their kills sometimes even before the predators themselves had even fed.
Acheulean tools represent a significant increase in complexity over Oldowan tools, as they required more time, effort, and skill to shape. Acheulean handaxes were not only worked on two sides, they shared a common shape, which required forethought and advanced planning by their makers. Homo erectus would have had a mental template for the desired outcome and, with practice, these tools were likely made quite quickly and could have been made by most individuals. While these tools were a significant step forward in tool production, they were not intended to be kept. Homo erectus discarded the tools after use and treated them as a disposable item.
In contrast, Neanderthal tools mark a significant innovation both in tool-making technique and their use. Known as , after the Le Moustier site in southwest France, the Neanderthal’s toolkit was truly that—a set of tools with specific applications and unique forms for each desired purpose. Mousterian tools were significantly smaller, thinner, and lighter than Acheulean handaxes and formed a true toolkit. The materials used for Mousterian tools were of higher quality, which allowed for both more precise toolmaking and tool reworking when the tools broke or dulled after frequent reuse. The use of higher-quality materials is also indicative of required forethought and planning to acquire them for tool manufacture. It is noteworthy that the Neanderthals, unlike Homo erectus, saved and reused their tools, rather than making new ones each time a tool was needed.

Mousterian tools are constructed in a very unique manner, utilizing the (Figure 11.8), named after the first finds of tools made with this technique, which were discovered in the Levallois-Perret suburb of Paris, France. The Levallois technique is a multistep process that requires preparing the core, or raw material, in a specific way that will yield flakes that are roughly uniform in dimension. The flakes are then turned into individual tools. The preparation of the core is akin to peeling a potato or carrot with a vegetable peeler—when peeling vegetables, you want to remove the skin in long, regular strokes, so that you are taking off the same amount of the vegetable all the way around. In the same way, the Levallois technique requires removing all edges of the , or outside surface of the raw material, in a circle before removing the lid. The flakes, which will eventually be turned into the individual tools, can then be removed from the core. The potential yield of tools from one core would be many, as seen in Figure 11.9, compared to all previous tool-making processes, in which one core yielded a single tool.

Neanderthal tools were used for a variety of purposes. They would have constructed a tool for each specific task they needed to complete, such as cutting, butchering, woodworking or antler working, and hide working. Additionally, because the Mousterian tools were lighter than previous stone tools, Neanderthals could , or attach the tool onto a handle, as the stone would not have been too heavy. Neanderthals attached small stone blades onto short wood or antler handles to make knives or other small weapons, as well as attached larger blades onto longer shafts to make spears. New research examining tar-covered stones and black lumps at several Neanderthal sites in Europe suggests that Neanderthals may have been making tar by distilling it from birch tree bark, which could have been used to glue the stone tool onto its handle. If Neanderthals were, in fact, manufacturing tar to act as glue, this would predate modern humans in Africa making tree resin or similar adhesives by nearly 100,000 years! While research on specific applications continues, from just this brief discussion, it should be clear that Neanderthal tool manufacturing was much more complex than previous tool-making efforts, requiring skill and patience to carry out.
With their more sophisticated suite of tools, Neanderthals were better armed for hunting than previous hominins and had very robust bodies with larger muscles. The animal remains in Neanderthal sites show that unlike earlier archaic Homo sapiens, Neanderthals were very effective hunters who were able to kill their own prey, rather than relying on scavenging. Oftentimes, this included very large animals like deer, horses, and bovids (relatives of the cow). In fact, isotopes from Neanderthal bones show that meat was a primary and significant component of their diet, similar to that seen in carnivores like wolves (Bocherens et al. 1999). Of course, Neanderthals’ diet varied according to the specific environment in which they lived, but according to Christoph Wiẞin and colleagues (2015), meat comprised up to 80% of their diet.
Though more sophisticated than the tools of earlier hominins, the Neanderthal spear was not the kind of weapon that would have been thrown; rather, it would have been used in a jabbing fashion (Churchill 1998; Kortlandt 2002). This may have required Neanderthals to hunt in groups rather than individually, and it almost certainly meant that they would have had to approach their prey quite closely. Remember, the animals living with Neanderthals were very large-bodied due to their adaptations to cold weather. In addition to large bovids, prey included ibex, seals, rabbits, and pigeons. Though red meat was a critical component of the Neanderthal diet, evidence shows that at times they also ate limpets, mussels, and pine nuts. Tartar examined from Neanderthal teeth in Iraq and Belgium reveal that plant material including wheat, barley, date palms, and tubers were also eaten by Neanderthals and were cooked to make them palatable.
While the new, close-range style of hunting used by Neanderthals was effective, it also had some major consequences. Many Neanderthal skeletons have been found with significant injuries, which could have caused paralysis or severely limited their mobility. Many of the injuries are to the head, neck, or upper-body. Thomas Berger and Erik Trinkaus (1995) conducted a statistical comparative analysis of Neanderthal injuries compared to those recorded in modern day workers’ compensation reports and found that the closest match was between Neanderthal injuries and those of rodeo workers. Rodeo professionals have a high rate of head and neck injuries that are similar to the Neanderthals’ injuries. What do Neanderthals and rodeo workers have in common? They were both getting very close to large, strong animals, and at times their encounters might have gone awry.
The extensive injuries sustained by Neanderthals are evident in many fossil remains. Shanidar 1 (Figure 11.10), an adult male found at the Shanidar site in northern Iraq and dating to 45,000 ya, has a lifetime of injuries recorded in his bones. Shanidar 1 sustained—and healed from—an injury to his face that would have likely caused blindness. His lower right arm was missing and his right humerus shows severe atrophy, likely due to disuse. This pattern has been interpreted to indicate a substantial injury that required or otherwise resulted in amputation or wasting away of the lower arm. Additionally, Shanidar 1 suffered from severe arthritis in his feet and bony growths in his inner ear that would have significantly impaired his hearing. He also exhibited extensive anterior tooth wear, matching the pattern of wear found among modern populations who use their teeth as a tool. Rather than an anomaly, the type of injuries evident in Shanidar 1 are similar to those found in many other Neanderthal fossils, revealing injuries likely sustained from hunting large mammals as well as demonstrating a long life of physical activity.
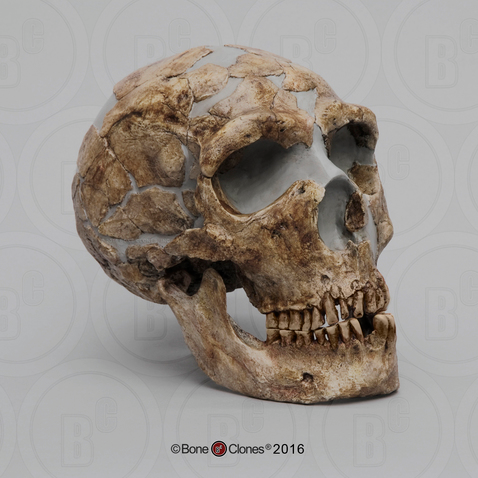

Figure 11.10 Like many other Neanderthals, Shanidar 1 has a lifetime of injuries recorded in his bones. Shanidar 1 sustained—and healed from—an injury to his face that would have likely caused blindness. His lower right arm was missing and his right humerus shows severe atrophy, likely due to disuse.
The pattern of injuries is as significant as the fact that Shanidar 1 and other injured Neanderthals often show evidence of having survived their severe injuries. One of the earliest known Neanderthal discoveries—the one on whom misinformed analysis shaped the stereotype of the species for nearly a century—is the La Chapelle-aux-Saints Neanderthal. The La Chapelle Neanderthal had a damaged eye orbit that likely caused blindness and suffered arthritis of the spine. He had also lost of most of his teeth, many of which he had lived without for so long that the mandibular and maxillary bones were partially reabsorbed due to lack of use. The La Chapelle Neanderthal was also thought to be at least in his mid-40s at death, an old age for the rough life of the Late Pleistocene, giving rise to his nickname, “the Old Man.” To have survived so long with so many injuries that obviously precluded successful large game hunting, he would have had to have been taken care of by others. Such caretaking behavior is also evident in the survival of other seriously injured Neanderthals, such as Shanidar 1. Long thought to be a hallmark human characteristic, taking care of the injured and elderly, to the extent of even preparing or pre-chewing food for those without teeth, indicates strong social ties among Neanderthals.
The care expressed in taking care of the sick or injured may have been expressed upon death as well. Full Neanderthal skeletons are not uncommon in the fossil record, and many of these skeletons were so well preserved due to having been placed in deliberate burials. These burials appear intentional, as the graves are dug down a bit, and the bodies found in the graves are in specific positions quite distinct from the natural position the body automatically goes into after death during rigor mortis. Neanderthal burials are often in a , or fetal position. Discoveries of pollen in a grave at the Shanidar site in the 1960s led scientists to think that perhaps Neanderthals had deliberately placed flowering plants in the grave, an indication of ritual ceremony or spirituality so common in modern humans. But future investigations have raised some doubt about this conclusion. The pollen may have been brought in by burrowing rodents. Claims of or other ornamentation in burials are similarly debated, although possible.
Some tantalizing evidence for symbolism, and debatably, ritual, is the frequent occurrence of natural pigments, such as (red) and manganese dioxide (black) in Neanderthal sites. Such pigments could have been used for art, like some of the spectacular cave paintings produced by modern humans who lived in this area after the Neanderthals. However, how these pigments were actually used by Neanderthals themselves is unclear, as there is very little evidence of art or paintings in Mousterian sites. One exception may be the recent discovery in Spain of a perforated shell that appears to be painted with an orange pigment, which may be some of the best evidence of Neanderthal art and jewelry. However, many pigments also have properties that make them good emulsifiers in adhesive (like for attaching a stone tool to a wooden handle) or useful in tanning hides. So the presence of pigment may or may not be associated with symbolic thought, but it at the very least shows a technological sophistication beyond that exhibited by earlier archaic hominins and clearly counters the old stereotypes of Neanderthals as dumb, thoughtless brutes.
The more recent time period in which Neanderthals lived and extensive excavations completed across Europe allow for a much more complete archaeological record from this time period. Additionally, the increased cultural complexity such as complex tools and ritual behaviors expressed by Neanderthals left a more detailed record than previous hominins. Intentional burials enhanced preservation of the dead and potentially associated ritual behaviors. Such evidence allows for a more complete and nuanced picture of this species.

Additional analyses are possible on many Neanderthal finds, due to increased preservation of bone, the amount of specimens that have been uncovered, and the recency in which Neanderthals lived. These additional studies include the examination of dental calculus and even DNA analysis. While limited, some samples of Neanderthal DNA have been successfully extracted and analyzed. Studies thus far have identified specific genetic markers that show some Neanderthals were light-skinned and probably red-haired with light eyes. Genetic analyses, different than the typical hominin reconstruction done with earlier species, allow scientists to further investigate soft tissue markers of Neanderthals and other more recent hominin species. These studies and Neanderthal cultural behavior have given scientists a wealth of information to study and offer striking conclusions regarding Neanderthal traits, their physical appearance, and their culture, as reflected in these artists’ reconstructions (Figure 11.11).
The Neanderthals’ more complicated behavior likely stems, in part, from their larger brains. Evidence shows that raw materials used by Neanderthals came from distances as far away as 100 km. This could indicate a variety of things regarding Neanderthal behavior, including a limited trade network with other Neanderthal groups, or simply a large area scoured by Neanderthals when collecting raw materials. Additionally, we know that Neanderthals lived in groups and may have relied on their group members for survival. Shanidar 1 and the “Old Man at La Chapelle” would have struggled to acquire and consume food on their own, strongly suggesting that they may have been assisted by relatives of other group members. In other nonhuman primates (like chimpanzees) and earlier hominins, injured individuals would have been left on their own, to either survive or perish.
The impressive cultural innovations and behavioral expansions seen in the Neanderthals would have required at least a basic form of communication in order to function, which suggests to many researchers that Neanderthals spoke. The challenge with this line of research is that speech, itself, of course is not preserved, so indirect evidence must be used to support this conclusion. It is thought that Neanderthals would have possessed some basic speech, as evidenced from a variety of sources, including throat anatomy and genetic evidence. There is only one bone in the human body that could demonstrate if a hominin was able to speak, or produce clear vocalizations like modern humans, and that is the hyoid, a U-shaped bone that is found in the throat and is associated with the ability to precisely control the vocal cords. Very few hyoid bones have been found in the archaeological record; however, a few have been uncovered in Neanderthal burials. The shape of the Neanderthal hyoid is nearly identical to that of modern humans, pointing to the likelihood that they had the same vocal capabilities as modern humans. Genetic evidence has been debated concerning the likelihood of speech. Geneticists have uncovered a possible mutation, the FOXP2 gene, that has been linked to the ability to speak and that both modern humans and Neanderthals possess. However, some scientists counter those findings, indicating that the study’s sample size was too small to make sweeping conclusions that the FOXP2 gene is what accounts for human or Neanderthal speech. Finally, scientists have also pointed to the increasingly complex cultural behavior of Neanderthals as a sign that symbolic communication, likely through speech, would have been the only way to pass down the skills needed to make, for example, a Levallois blade or to position a body correctly for intentional burial.
Neanderthal Intelligence
One of the enduring questions about Neanderthals centers on their intelligence, specifically in comparison to modern humans. Brain volume indicates that Neanderthals certainly had a large brain, but it continues to be debated if Neanderthals were of equal intelligence to modern humans. Brain volume, cultural complexity, tool use, and compassion toward their kind all point to an increase in intellect among Neanderthals when compared to previous hominins.
However, there have been several studies that seem to indicate that while Neanderthals did have a large brain volume and were far more advanced than their previous relatives, they may not have been nearly as intelligent as or may have even lacked the intellectual abilities possessed by modern humans. Euluned Pearce and colleagues, from the University of Oxford, noted that based on cranial endocasts, the frontal lobe of Neanderthals and modern humans are almost identical. However, Neanderthal faces and other cranial features were larger. Neanderthals possessed larger eye sockets, and the larger eyes they held would have helped Neanderthals see in the low light levels common for the latitudes at which they lived. Because of the larger eye sockets, the visual cortex—the portion of the brain involved in processing visual information—would have had to have been enlarged, as well. This would have left Neanderthals with less neural tissue for other components of the brain, including those that would have aided them in dealing with expansive social networks, one of the differences that it has been suggested existed between Neanderthals and modern humans.
New research is suggesting additional differences between Neanderthal brains and our own. Research being conducted by geneticist John Blangero and his team from the Texas Biomedical Research Institute are examining genes involved in certain diseases among modern populations. His team has also looked at brain structure and function. Comparing data from the Neanderthal genome against MRI data from his modern study participants, Blangero and his team discovered that some Neanderthal brain components were very different, and smaller, than those in the modern sample. These areas include decreased gray matter surface area, a smaller amygdala, and less white matter. These three regions are important in the processing of information and controlling emotion and motivation, as well as overall brain connectivity. In short, as Blangero stated at the annual meeting of the American Association of Physical Anthropologists in 2014, “Neanderthals were certainly cognitively adept,” although their specific abilities may have differed from modern humans’ in key areas (qtd. in Wong 2015), a point echoed in other recent genetic studies comparing Neanderthal and anatomically modern human brains (el-Showk 2019).
Finally, scientists are fairly certain that Neanderthal brain development after birth was not the same as that of modern humans. After birth, anatomically modern Homo sapiens babies go through a critical period of brain expansion and cognitive development. It appears that Neanderthal babies’ brains did not follow the same developmental pattern. This could also be related to the length of the period of childhood. Modern humans enjoy an extended period of childhood, which, among many things, allows children to engage in imaginative play and develop creativity. Child development studies indicate that children who have extended, undirected play opportunities will be better off academically and socially later in life than their peers who had less play opportunities. Based on their anatomical developments, it appears that Neanderthals had a limited childhood. It has been suggested that this limited time for play and developing a creative mind might have limited adult creativity and how successful they were as a species, in the long run.
The exact nature of Neanderthal intelligence remains under investigation, however. Some studies disagree with the idea that Neanderthal intelligence had limitations compared to our own, noting that there is extensive evidence showing that Neanderthals displayed limb asymmetry. Their tools also have wear marks indicating that they were hand-dominant. It has been established that favoring the right hand is one key marker between modern humans and chimpanzees, and that handedness is likely also related to language development, in the form of bilateral brain development. That Neanderthals likely were hand-dominant as well suggests that they at least had many of the preconditions for human speech and likely experienced similar bilateral brain development to our own.
In addition to cut marks on animal bones, there are marks on Neanderthal teeth that demonstrate hand dominance. Neanderthal upper incisors, or front teeth, show not only wear from using their teeth when preparing hides or cordage but also cut marks that were created by using their teeth as a third limb when eating. The “stuff-and-cut method,” noted by David Frayer, would have seen the Neanderthal hold a piece of meat in their teeth and pull it taut with one hand, and then, using their other hand, their dominant one, cut meat off the slab. When looking at 17 Neanderthals and their tooth wear, only two do not show markings made by a right-hand dominant individual. This research suggests another similarity between Neanderthal and modern human brains and their associated intelligence.
THE MIDDLE STONE AGE: NEANDERTHAL CONTEMPORARIES IN AFRICA
While Neanderthals made their home on and adapted to the European and Asian continents, evidence of fossil humans in Africa show they were also adapting to their local environments. These populations in Africa exhibit many more similarities to modern humans than Neanderthals, as well as overall evolutionary success. While the African fossil sample size is smaller and more fragmentary than the number of Neanderthal specimens across Europe and Asia, the African sample is interesting in that it represents a longer time period and larger geographical area. This group of fossils, often represented under the name of “Middle Stone Age,” or MSA, dates to between 300,000 and 30,000 years ago across the entire continent of Africa. As with archaic Homo sapiens, there is much variability seen in this African set of fossils. There are also a few key consistent elements: none of them exhibit Neanderthal skeletal features; instead, they demonstrate features that are increasingly consistent with anatomically modern Homo sapiens.
Similarities to Neanderthals and MSA contemporaries in Africa are seen, however, in their behavioral adaptations, including stone tools and other cultural elements. The tools associated with the specimens living in Africa during this time period are, like their physical features, varied. In some parts of Africa, namely Northern Africa, stone tools from this time so closely resemble Neanderthal tools that they are classified as Mousterian. In sub-Saharan Africa, the stone tools associated with these specimens are labeled as Middle Stone Age, or MSA. Some scholars argue that these could also be a type of Mousterian tools, but they are still typically subdivided based on geographical location.
Recall that Mousterian tools were much more advanced than their Acheulean predecessors in terms of how the stone tools were manufactured, the quality of the stones used, and the ultimate use of the stone tools that were made. In addition, recent evidence suggests that MSA tools may also have been heat treated—to improve the quality of the stone tool produced. Evidence for heat treating is seen not only through advanced analysis of the tool itself but also through the residue of fires from this time period. Fire residues show a shift over time from small, short fires fueled by grasses (probably intended for cooking) to larger, more intensive fires that required the exploitation of dry wood, exactly the type of fire that would have been needed for heat treating stone tools.
Other cultural elements seen with specimens dating to the MSA include use of marine (sea-based) resources for their diet, manufacture of bone tools, use of adhesive and compound tools (e.g., hafted tools), shell bead production, engraving, use of pigments (such as ochre), and other more advanced tool-making technology (e.g., microlithics). While many of these cultural elements are also seen to a limited extent among Neanderthals, many of the developments at MSA sites are far more complex than what is demonstrated with Neanderthal sites. Several explanations have been posited to explain this expansion of cultural complexity. It has been suggested that MSA cultural expansion was a response to climate change. It has also been suggested that perhaps the MSA cultural expansion was due to an increased use of language, which triggered increased symbolic thought. Others have suggested that the cultural expansion of the MSA was due to the increase of marine resources in their diet, which included more fatty acids and may have aided their cognitive development. Still others have suggested that the increased cultural complexity was due to an increase in competition and interaction among groups, which spurred competition to innovate with increased cultural complexity. Recent studies suggest that perhaps the best explanation for the marked cultural complexity and diversity demonstrated by MSA cultural artifacts is best explained by the simple fact that they lived in diverse habitats. This would have necessitated a unique set of cultural adaptations for each habitat type (for example, specialized marine tools would have been needed along coastal sites but not at inland locations). Simply put, the most useful adaptation of MSA was their flexibility of behavior and adaptability to their local environment. As noted previously in this chapter, flexibility of behavior and physical traits, rather than specialization, seems to be a feature that was favored in hominin evolution at this time.
WHERE DID THEY GO? THE END OF NEANDERTHALS
While MSA were increasingly successful and ultimately transitioned into modern Homo sapiens, Neanderthals disappear from the fossil record by around 35,000 years ago. The main question that lingers, however, is what happened to them. We know, based on genetics, that modern humans come largely from the modern people who occupied Africa around 300,000 to 100,000 years ago, at the same time that Neanderthals were living in the icy portions of northern Europe and Asia. Modern humans expanded out of Africa around 60,000 to 40,000 years ago, rapidly entering areas of Europe and Asia inhabited by Neanderthals and likely other populations of archaic hominins. Despite intense interest and speculation in fictional works about possible interactions between these two groups, there is very little direct evidence of either peaceful coexistence or aggressive encounters. It is clear, though, that these two closely related hominins shared Europe for thousands of years, and recent DNA evidence suggests that they at least occasionally interbred. Geneticists have found traces of Neanderthal DNA (1% to 4%) in modern humans of European and Asian descent that is not present in modern humans from Africa. This is generally interpreted as indicating limited regional interbreeding with Neanderthals. Interestingly, while some studies suggest interbreeding was often problematic for their offspring, gene flow from Neanderthals may have benefited modern Homo sapiens. David Enard and Dmitri Petrov (2018) compared sequenced Neanderthal and modern human DNA, observing that the portions of Neanderthal DNA in modern humans of European descent seem to confer defense against viral pathogens that they would have been exposed to as they moved out of Africa.
While some interbreeding likely occurred, as a whole, Neanderthals did not survive. What is the cause for their extinction? This question has fascinated many researchers and several possibilities have been suggested that deserve some exploration. Possibilities include the following:
- The climate began changing considerably from the height of Neanderthal expansion. At the time that Neanderthals were disappearing from the fossil record, the climate went through both cooling and warming periods—each of which posed challenges for Neanderthal survival (Defleur and Desclaux 2019; Staubwasser et al. 2018). It has been argued that as temperatures warmed, large-bodied animals, well adapted to cold weather, moved farther north to find colder environments or face extinction themselves. A shifting resource base could have been problematic for continued Neanderthal existence, especially as additional humans, in the form of modern Homo sapiens, began to appear in Europe and were competing with them for a smaller pool of available resources.
- It has been suggested that the eruption of a European volcano 40,000 years ago could have put a strain on available plant resources (Golovanova et al. 2010). The eruption would have greatly affected local microclimates, reducing the overall temperature enough to alter the growing season.
- Possible differences in cognitive development may have limited Neanderthals in terms of their creative problem solving. It has been argued that as much as they were biologically specialized for their environment, the nature of their intelligence might not have offered them the creative problem solving skills to rethink their cultural adaptations and innovate ways to adapt their culture when faced with a changing environment (Pearce, Stringer, and Dunbar 2013).
- There is evidence that suggests reproduction may have posed challenges for Neanderthals. Childbirth was thought to have been at least as difficult for female Neanderthals as anatomically modern Homo sapiens (Weaver and Hublin 2009). Female Neanderthals may have become sexually mature at an older age, even older than modern humans. This delayed maturation could have kept the Neanderthal population size small. A recent study has further suggested that male Neanderthals might have had a genetic marker that would have had negative impacts on the longevity of the Neanderthal population (Mendez et al. 2016).
- We would be remiss if we did not point out that the end of Neanderthal existence also overlaps with modern human expansion into northern Europe and Asia. There is no conclusive direct evidence to indicate that Neanderthals and modern humans lived peacefully side by side, nor that they engaged in brutal warfare, but by studying modern societies and the tendencies of modern humans, it has been suggested that modern humans may not have warmly embraced their close but slightly odd-looking cousins when they first encountered them (Churchill et al. 2009). Competition for resources may have been the cause of the Neanderthals’ decline (Gilpin, Feldman, and Aoki 2016). It is also completely possible that modern humans gave Neanderthals diseases to which they previously had little to no exposure, causing a mass population decline similar to what happened when the invading Spanish wiped out the Mayan and Aztec populations (Houldcroft and Underdown 2016). Estimates of energy expenditures suggest Neanderthals had slightly higher caloric needs than modern humans (Venner 2018). When competing for similar resources, the slightly greater efficiency of modern humans might have helped them experience greater success in the face of competition—at a cost to Neanderthals.
- Finally, less dramatically yet still significantly, even a small but continuous decrease in fertility would have been enough to result in the extinction of Neanderthals (Degioanni et al. 2019).
As Neanderthal populations were fairly small to begin with (estimated between 5,000 and 70,000 individuals) (Bocquet-Appel and Degioanni 2013), one or a combination of these factors could have easily led to their demise. As more research is conducted, we will likely get a better picture of exactly what led to Neanderthal extinction.
DENISOVANS
While Neanderthals represent one regionally adapted branch of the archaic Homo sapiens family tree, recent discoveries in Siberia and the Tibetan Plateau have surprised paleoanthropologists by revealing yet another population that was contemporary with archaic Homo sapiens, Neanderthals, and modern Homo sapiens. The genetic analysis of a child’s finger bone and an adult upper third molar from Denisova Cave in the Altai Mountains in Siberia by a team including Svante Pääbo shocked even the researchers when they discovered that the mitochondrial and nuclear DNA sequences revealed distinct genetic differences from all known archaic populations. Dubbed “Denisovans” after the cave in which the bones were found, this population is more closely related to Neanderthals than modern humans, suggesting the two groups shared an ancestor who split from modern humans first, then the Neanderthal-Denisovan line diverged more recently.
Denisovans share up to 5% of their DNA with modern Melanesians, aboriginal Australians, and Polynesians, and 0.2% of their DNA with other modern Asian populations and Native Americans. Additional studies have suggested two separate instances of interbreeding between humans and Denisovans, whom researchers have yet to classify as a separate species, pending additional information.
Genetic analysis reveals that Denisovans potentially had at least three populations and had genetic adaptations for life at high altitudes, preventing them from developing altitude sickness and hypoxia. Recent publications also suggest that Denisovans shared these genetic adaptations with modern Tibetans through interbreeding 30,000 to 40,000 years ago. Stone tools similar to those found in Siberia have also been found in the Tibetan plateau, suggesting the possibility that Denisovans could have inhabited this extreme environment where the average annual temperature is close to 0℃ and the altitude is more than a kilometer (about 4,000 feet) above sea level. Research continues on this population and other archaic populations in the hopes of discovering more DNA evidence that can confirm current hypotheses and clarify our understanding of the complex interactions of archaic groups.
To stay up to date with new discoveries, consider following organizations such as the Smithsonian’s Human Origins Program on social media (https://www.facebook.com/smithsonian.humanorigins/).
MODELS OF MODERN HUMAN DISTRIBUTION
There has been much debate in anthropological circles concerning the origin of modern humans and their relationship with other hominin populations. Three competing models have been developed and seek to explain the fossil evidence and what it indicates for modeling human origins.
The first model, the Out-of-Africa Hypothesis, states that modern humans originated in Africa, replacing archaic populations found elsewhere in the Old World. Theorists including Christopher Stringer (1996) argue that each archaic population comprised a separate species, making interbreeding between populations impossible. Admixture resulting from gene flow would not have been possible according to this model.
The second, called the Multiregional Continuity Hypothesis, states that modern Homo sapiens are directly derived from Homo erectus and evolved in place after Homo erectus left Africa and populated areas in Asia and Europe. Milford Wolpoff argues that interbreeding between regions and across regional boundaries contributed to gene flow that maintained Homo sapiens as a single species throughout the Old World, despite regional variation.
The third model, dubbed the Assimilation Hypothesis, draws from the strengths of both previous models, attempting to recognize some of the evidence that was not previously addressed and blending the fossil and DNA evidence together into one cohesive view. In this model, modern humans originated in Africa, spreading outward into Asia and Europe and interbreeding with more archaic forms they encountered along the way. For example, while the Out-of-Africa model argues that interbreeding would have been impossible, many fossils have been found with what appear to be a mixture of archaic and more modern traits, suggesting interbreeding between populations, such as Neanderthals and modern humans. DNA evidence increasingly also suggests that, while limited, interbreeding between modern Homo sapiens and Neanderthals or modern Homo sapiens and Denisovans occured in at least three instances. While this is more interbreeding than allowed under the Out-of-Africa Hypothesis, it is considerably less than modeled in the Multiregional Continuity Hypothesis. The Assimilation Hypothesis, argued by Eric Trinkaus (2006, 2007) and others, represents an attempt to incorporate all lines of evidence, although new research will tell whether it can capture the full complexity revealed in the next generation of hominin studies, such as that revealed by ancient DNA.
SPECIAL TOPIC: ANCIENT DNA
Robyn Humphries, MSc., University of Cape Town
Ancient DNA has provided us with new insights into our evolutionary history that cannot be garnered from the fossil record. It has also assisted with the discovery of the new hominin species the Denisovans, for which little fossil evidence is available. It has helped us better understand the evolution of Neanderthals, Denisovans, and modern humans. Through genomic data and the use of population genetics, we have been able to make some inferences about Neanderthal and Denisovan population structure and relationships within these populations as well as between different groups of hominins. It has also helped to answer some very important questions about what happened when modern humans migrated out of Africa and encountered these European/Asian hominins. Two theories dominated the debate regarding the evolution of modern humans: the multiregional theory and the Out-of-Africa theory. Though it was clear—based on a plethora of evidence—that modern humans evolved in Africa, what happened when our ancestors migrated out of Africa was still questioned. Ancient DNA (aDNA) helped answer this question, indicating that modern humans interacted with other archaic hominins such as Neanderthals and Denisovans. We will discuss all the above in this section.
Sequencing Ancient Genomes
The first successful sequencing of aDNA from an archaic hominin took place in 1997 with the sequencing of mitochondrial DNA (mtDNA) from the Neanderthal-type specimen from Feldhofer Cave. Sequencing of a portion of the mitochondrial genome provided molecular evidence that Neanderthals belonged in a clade separate from modern humans and that they were four times more different from modern humans than modern humans were from each other based on mtDNA data. mtDNA is ideal for sequencing from fossil material because of the abundance of mtDNA when compared to nuclear DNA.
Sequencing of nuclear DNA would not occur until more than ten years later. The first nuclear genomic sequence representing Neanderthals was produced by sequencing three individuals and using their sequences to create a composite draft Neanderthal genome in 2010. The first high-coverage sequence of a single Neanderthal was that of a female Neanderthal who lived in Siberia, which was published in 2014, followed by another high-coverage sequence from a female Neanderthal whose remains were found in the Vidja cave in Croatia, which was published in 2017. are produced when the genome has been sequenced multiple times. This is to ensure that the sequences obtained are a true reflection of the genomic sequence and not due to errors that occur during the process of sequencing. If you have many sequences from the same region and there is one sequence that has a slight difference while the other copies are all the same, it is easier to identify the variant as an error.
Collecting and Sequencing aDNA
Ancient DNA can be collected from many different sources including soft tissue such as skin and muscle, hair, paleo feces, soils, and sediments. However, in the case of ancient hominins, they are often collected from bone and teeth. When collecting aDNA, usually around 100 mg to 500 mg of bone powder needs to be collected. Because extraction of aDNA requires destruction of part of the bone, and the morphology of the skeletal element might be informative, care needs to be taken when deciding which part of the bone is sampled. It is advised that multiple samples be taken so that sequencing is repeated to show reproducibility of results. Contamination is an important consideration when it comes to sequencing aDNA; thus, it is best that samples that are used had minimal handling before extraction of DNA.

It has taken a lot of time and much trial and error to sequence these ancient genomes because of the fragility of DNA. When sequencing ancient DNA, it is important to consider that aDNA sequences are usually short due to degradation, there are very few copies of the . Endogenous aDNA is the DNA that comes from the bone and was present in the tissue before decomposition of the body and before the introduction of DNA from other sources, such as microbes or contamination from modern humans, which is known as (Figure 11.12).
There are also modifications that occur to aDNA that are a result of chemical reactions known as deamination. results in Cytosine (C) to Thymine (T) conversions, which are mostly at the 5’ end ()of the DNA fragment. This in turn results in Guanine (G) to Adenine (A) substitutions on the 3’ end ()of the DNA fragment. Thus, there are sequence changes in aDNA that might not reflect the original hominin sequence. These changes can be helpful when differentiating between aDNA and modern human DNA contamination. The environment in which the DNA is preserved also plays a significant role. DNA preserves well in cold conditions such as permafrost, which extends the lifespan of DNA significantly. aDNA has also been recovered from material found in drier environments under special conditions. Factors such as water percolation, salinity, pH, and microbial growth all affect the preservation of aDNA.
In extraction of DNA from modern samples where DNA is still intact, the DNA strands are usually long and this is ideal for sequencing. However, aDNA samples are often composed of small fragments of DNA, usually 100 bp to 300 bp long. Initially this posed a big problem with usual PCR procedures used to sequence DNA. This changed with the advent of high throughput sequencing, which has revolutionized sequencing the genomes of ancient hominins. High throughput sequencing allows for the parallel sequencing of many fragments of DNA in one reaction. It also doesn’t require any knowledge of the target sequence. Thus, we can sequence as much of the available aDNA as possible. Because the high throughput sequencing method does not discriminate between endogenous aDNA from hominins and contamination from modern humans and microbial DNA, it is important to either ensure that there is as little contamination as possible or create methods that allow for differentiation between modern human sequences and ancient hominin sequences. Both methods have been used when sequencing hominin aDNA.
The Discovery of the Denisovans
The Denisovans are named after the cave in which they were discovered, the Denisovan Cave in the Altai Region of Siberia. Denisovans were initially identified as a distinct group based on analysis of mtDNA sequences indicating that they had haplotypes outside the range of variation of modern humans and Neanderthals. A is a set of genetic variants located on a single stretch of the genome. This unique combination of variants on a stretch of the genome can be used to differentiate groups who will have different combinations of variants. Some haplotypes may be more similar to one another. The more similar two haplotypes are, the more closely related they are. Dubbed lineage X, the mtDNA sequence showed that Denisovans diverged from modern humans and Neanderthals at around 1 million years ago (mya). The subsequent high-coverage sequence of a Denisovan 3 nuclear genome showed that Denisovans are a sister group to Neanderthals and thus more closely related than indicated by the mtDNA data.
The mtDNA and nuclear DNA provided conflicting data regarding the relationships between Denisovans and Neanderthals. Because mtDNA and nuclear DNA have different patterns of inheritance, they can paint different pictures about the relationships between two groups when used to construct phylogenies. The Denisovans are thought to have a mtDNA sequence that is derived from an ancient hominin group that hybridized with Denisovans and introduced the mtDNA sequence.
Sequences are also available for three other Denisovans, Denisovan 2, 4, and 8. aDNA sequences have been used to estimate the ages of the Denisovans. Using a combination of usual dating methods (such as radio carbon dating and uranium dating) as well as genetic data, it has been determined that Denisovans occupied the Denisovan cave from around 195 kya to 52 kya to 76 kya. DNA can assist with dating because younger sequences will have accumulated more sequence changes from the putative common ancestral sequence than older samples. This is because younger sequences would have had more time over which changes to the DNA sequence through mutation could occur. Thus, it is possible to conclude based on sequence data that Denisovan 2 is 54.2 kya to 99.4 kya older than Denisovan 3 and 20.6 kya to 37.7 kya older than Denisovan 8. Molecular data indicates that Neanderthals and Denisovans separated between 381 kya and 473 kya and that the branch leading to Denisovans and modern humans diverged around 800 kya. Denisovans are also more closely related to another set of fossils found in the cave Sima de los Huesos dated to 480 kya. Thus, the split between Neanderthals and Denisovans must have occurred before 480 kya.
What Can We Learn about Population Structure of the Neanderthals and Denisovans from aDNA
Ancient DNA has helped us understand the demographics of Neanderthals and Denisovans and make inferences about population size and history. The genomic data from Neanderthals indicates that their population was small toward the end of their existence. This is supported by three lines of evidence.
The first is by using methods. This is the process used to determine which population dynamics in the past are most likely to give rise to the genetic sequences we have, allowing us to use genetic sequences to estimate population genetic parameters in the past. It can be used to understand recombination, population subdivision, and variable population size.
The second indicator that Neanderthals and Denisovans had smaller population size is that these groups carried many deleterious genomic variants. Genomic variants are considered deleterious when they are found in protein-coding regions of the genome and the change in genomic sequence translates to a change in amino acid sequence of the protein. Changes in amino acid at a certain section in the protein could affect the functioning of the protein—these types of changes in genomic sequence are known as non-synonymous mutations. also occur in protein-coding regions of the genome, but the amino acid sequence does not change because of changes in the genomic sequence. Changes in amino acid and subsequent protein sequence can change the protein function and thus are more likely to be deleterious and weeded out by natural selection. The ratio of synonymous to can give you an indicator of whether there are more deleterious variants than expected. Denisovans and Neanderthals have a higher ratio of non-synonymous to synonymous mutations when compared to contemporary modern human populations. This is an indicator of a small population size, because if the population were larger, natural selection would have acted on these deleterious variants and weeded them out.
A third indicator of small population size is that the Neanderthals sequenced thus far have low levels of , a measure of how many genes within a genome are made up of more than one variant. Each individual has two copies of the same gene: one is inherited from their mother and the other from their father. Variations of the same gene are known as , which are versions of the same gene with h different sequences. If the alleles inherited from both parents are the same, the individual is homozygous for that gene; if the alleles inherited are different, the individual is heterozygous for that gene. Heterozygosity is measured by looking at how many times you happen to find two different alleles within a certain stretch of DNA. When you find many regions on the genome with different alleles, there is a high level of heterozygosity. When you find very few positions where there are two different alleles, this results in a low level of heterozygosity.
Ancient Neanderthal genomes also revealed that there were consanguineous relations between Neanderthals. One Neanderthal female is thought to be the offspring of relations between either half-siblings, an uncle/aunt and niece/nephew, or a grandfather/grandmother and grandson/granddaughter. This was determined by looking at the stretches of in her genome that were longer than expected and could not be explained by small population size alone.
Denisovans also had low levels of heterozygosity indicating a smaller population size. However, there is no indication yet of inbreeding among the Denisovans, as none of the individuals sequenced thus far show long stretches of homozygosity. Thus, both Denisovans and Neanderthals had small populations size and low levels of genetic diversity when compared to modern humans.
How Sequencing Archaic Genomes Can Help Understand Our Own Unique Evolutionary Trajectory as Modern Humans
Not only did the sequencing of archaic genomes allow us to learn more about Neanderthals and Denisovans, it gave us important insights into our own evolution. Previously the human genome could only be compared to our closest living relatives, the great apes, which helped us identify unique derived genomic changes that occurred in humans since our split from the last common ancestor between chimps and humans. Neanderthal and Denisovan genomes provided another set of comparative samples that could help us identify changes that were unique to modern humans occurring after our split from the last common ancestor with Neanderthals/Denisovans. We now have an opportunity to identify genetic variants that may have contributed to our success as a species.
Hybridization between Hominin Groups
Ultimately aDNA provides us with great insight into interactions between modern humans migrating out of Africa and other hominins that evolved in Europe and Asia. There was speculation that hybridization occurred due to the intermediate morphology of some fossil remains. The following hypothesis was tested: if hybridization between modern humans and Neanderthals occurred, Neanderthals would have more shared genomic variants with some modern human populations than with others. If this was true, hybridization between Neanderthals and humans happened. This comparison showed that Neanderthals shared more genomic variants with Europeans and Asians than with the African individuals. This difference in relatedness was significant. This indicated that there had been hybridization between Neanderthals and modern humans.
From the genetic data, we know that different groups have different amounts of Neanderthal and Denisovan contributions. For example, Europeans have a smaller proportion of Neanderthal-derived genes than East Asians. Thus, there was more admixture into ancestral East Asian populations than into ancestral European populations. This is unexpected because Neanderthals fossils are mostly found in West Asia and Europe. Oceanians (Melanesians, Australian aborigines, and other Southeast Asian islanders) have a higher proportion of their DNA derived from Denisovans. These populations also have longer stretches of Denisovan DNA. Since DNA in chromosomes get exchanged and “break apart” between each generation (in the process known as genetic recombination), this implies that the admixture event between the Denisovan and human ancestors of these populations is more recent than the admixture events between Neanderthals and modern humans. Genomic recombination breaks down introgressed regions (inherited from different species or taxon) into smaller segments in every successive generation, thus longer stretches of introgressed DNA indicates that hybridization occurred more recently.
Initially, some researchers believed that populations outside of Africa had higher proportions of Neanderthal DNA due to population substructure, which existed in the ancestral population before the split between modern humans and Neanderthals. According to these researchers, Eurasian populations retained these ancient sequences by chance, through genetic drift. These would be shared derived genomic sequences between Neanderthals and modern humans outside of Africa. However, studies of the shared regions indicate that these genomic regions are most likely the result of introgression, which is the transfer of genetic information from one species to another because of hybridization between them and repeated backcrossing.
Divergence time is important for determining whether shared sequences are a result of introgression or more ancient substructure. is a measure of how long two sequences have been changing independently. It is measured by looking at how many differences there are between the two sequences. The longer the two sequences have been changing independently, the more differences they will accumulate, which will result in a longer divergence time. By measuring the divergence time between the introgressed regions in modern human genomes and the Neanderthal sequences, researchers can calculate that the shared sequences are recent as well as date to when the two taxa made secondary contact. This is also well after the initial population split between modern humans and Neanderthals occurred. If they were shared derived genomic sequences, then we would expect longer divergence times between the introgressed Neanderthal genomic sequences in modern humans and the Neanderthal genome. Hybridization has occurred between hominins at different times over the last 100 kya as shown in Figure 11.14.
The Neanderthal and Denisovan genomes would provide definitive proof that there was interaction and interbreeding between humans, Neanderthals, and Denisovans around 44 to 55 kya, with data suggesting that there was an admixture event as far back as 100 kya. There has been gene flow from Neanderthals and Denisovans into modern human populations, between Neanderthals and Denisovans, and from modern humans into Neanderthals.
Because of the climate in Africa, it has been difficult or impossible due to fossilization to extract aDNA from African fossil remains. However, analysis of genomes of modern African populations indicate that there was admixture between modern humans and other hominins within Africa (Figure 11.13). Thus, hybridization is an important part of human evolution and has affected our evolution within and outside of Africa.

The oldest modern human that has been sequenced Ust’-Ishim is from Europe and is dated to around 49 kya. He had a similar amount of Neanderthal-derived genes as modern humans from outside of Africa. Analysis of the genome indicated that the hybridization event resulting in the introgression occurred 50 ky before. The fact that the Ust’-Ishim modern human had longer tracts of Neanderthal-derived DNA than contemporary populations lends support to the idea that Neanderthal-derived DNA in modern humans is due to hybridization. Contemporary modern humans have shorter stretches of Neanderthal-derived genes because there has been a longer period over which the Neanderthal segments of DNA could be broken down by .
Thus, there are multiple lines of evidence supporting hybridization between modern humans and Neanderthals/Denisovans. This includes shorter divergence times between introgressed regions in modern-human and Neanderthal sequences, older modern-human sequences having longer tracts of Neanderthal-derived genes and, as discussed below, the sequencing of confirmed hybrids.
Confirmed Fossil Hybrids
When discussing hybrids, there are some important terms to understand. A first-generation hybrid is called an F1 hybrid; it is the direct offspring of two lineages that have been evolving independently over an extended period. A second-generation hybrid (F2) would be the offspring of two F1 hybrids. A backcrossed individual is the result of an F1 or F2 hybrid mating with an individual from one of the parental populations. An example of a backcross would be when a Neanderthal-human hybrid produces offspring with a human; their offspring would be considered a first-generation backcrossed hybrid (B1). Sequencing of aDNA from fossil material has further confirmed that hybridization between different hominins has occurred, supporting the introgression data from recent populations.
The sequencing of Oase 1, a suspected hybrid based on skeletal morphology (what the fossil looked like), showed that it had a Neanderthal ancestor as recently as six to eight generations back. He would thus be considered a backcrossed individual. The recent sequencing of a 13-year-old Denisovan female showed that she was the F1 hybrid offspring of a Neanderthal mother (from whom she inherited Neanderthal mtDNA) and a Denisovan father. She was confirmed to be an F1 hybrid because approximately 50% of her genome was derived from a Neanderthal and 50% from a Denisovan.
These are only two examples of individuals who are confirmed hybrids. Many other remains show some indication of gene flow between hominins.
Neanderthal- and Denisovan-Derived DNA in Modern Genomes
There is variation in how much of the Neanderthal genome is represented in the modern human population. Individuals outside of Africa usually have 1% to 2 % of their genome derived from Neanderthals. Approximately 30% of the Neanderthal genome is represented in modern human genomes. Asian populations usually have a higher proportion of their genome derived from Neanderthals when compared to modern European populations. Additionally, the sequencing of the Denisovan genome indicates that they interacted with the ancestors of modern Oceanic populations. Thus, oceanic populations have around 5% to 6% of their genome derived from Denisovans. There is also evidence that different Denisovans populations may have contributed to Oceanians and East Asians. The available Denisovan sequences are more similar to the Denisovan introgressed genes found in East Asian populations.
have signatures that allow us to identify them and differentiate them from parts of the genome that are not introgressed. Some of the things to look for when determining if a segment of the genome is introgressed include the following. First, how closely does the segment you are looking at match the Neanderthal/Denisovan sequence compared to contemporary modern human sequences from Africa? If the sequence is more similar to the Neanderthal sequence (i.e., it has less sequence differences from the Neanderthal than the African modern human), it is likely that it is derived from a Neanderthal). Second, what is the divergence time between the allele and the same allele in a Neanderthal? If it is shorter than the divergence time between humans and Neanderthal, then the gene is most likely introgressed. This is expected because if the divergence time is after the split between modern humans and Neanderthals, the most likely explanation for a shorter divergence time is introgression. An example of this can be seen in Figure 11.14. And, third and finally, you need to look at whether the allele is found at higher frequencies in populations outside of Africa.

What Can We Learn about the Process of Hybridization from Ancient DNA?
Ancient DNA has also allowed us to make certain inferences about the process of hybridization between modern humans and Neanderthals/Denisovans. From looking at the genomes of modern humans, we can see that there are regions of the genome with no Neanderthal and Denisovan genomic variants. These are known as Neanderthal or Denisovan introgression deserts. There are also overlaps between regions in the human genome that are Neanderthal and Denisovan deserts, which might indicate that there were genomic incompatibilities between modern humans and these groups, resulting in those genes being selected against on the modern human genome background. This resulted in strong negative selection against these genomic variants in subsequent generations of hybrids and backcrossed individuals.
We can also infer that hybridization may itself have been a barrier to gene flow because there is a significant reduction in introgression on the X chromosome compared to the other chromosomes. There is also a reduction of introgressed genes around genes that are disproportionately expressed in the testes when compared to other tissue groups. This could indicate that hybridization between modern humans and Neanderthals may have resulted in male hybrid infertility.
Hybridization and Modern Human Evolution
Hybridization provided adaptive advantage to modern humans migrating out of Africa by providing them with advantages in genetic variation. Neanderthals and Denisovans had spent hundreds of thousands of years adapting to the European and Asian environments and thus had genetic variants favorable for inhabiting those regions. Through hybridization, humans were able to acquire favorable genomic variants already selected for, and these variants could rapidly spread through the population. This allowed for faster adaptation because acquiring new variation through mutation alone is much slower and less likely to spread through the population. Some of the adaptive genes that were important include genes associated with immunity, adapting to new diets, adapting to new altitudes as well as genes involved in skin color and hair traits were introgressed. An excellent example of this would be a variant of the EPAS1 gene found at high frequencies in Tibetans, thought to be important for living at high altitudes. This variant of EPAS1 has been shown to be an introgressed gene from Denisovans.
The Future of Genetic Studies
We are continuing to learn how introgressed genes affect modern humans. Combining phenotypic and genetic information Neanderthal derived genes have been associated with diverse traits such as the skin’s sensitivity to the sun to excessive blood clotting by certain individuals. Interesting research has also shown that introgressed alleles might produce different gene expression profiles when compared to non-introgressed alleles. However, there is a lot of research that needs to be done to fully understand the effects of introgression on modern populations and how it might have assisted modern humans who migrated out of Africa.
It has also been possible to extract DNA from sediments. However, extractions from sediments will result in extraction of DNA from multiple organisms. To extract the hominin sequences, we will need to use the known sequence information. Known sequences will also assist with differentiating between which hominins are represented in the sediment. This could assist with identifying changes in populations across time. The availability of more Neanderthal and Denisovan samples will also help us understand which genetic changes were fixed and defined these populations.
HOW DO THESE FIT IN? HOMO NALEDI AND HOMO FLORESIENSIS
Relatively recently, some fossils have been unearthed that have significantly challenged our understanding of the hominin lineage. The fossils of Homo naledi and Homo floresiensis are significant for several reasons but are mostly known for how they don’t fit the previously held patterns of hominin evolution. We’ll examine present information about both of these fossils, and we ask that you consider the wealth of evidence presented in this chapter and in others to draw your own conclusions regarding the significance and placement of these two unusual fossil species in the hominin lineage.
Homo naledi

Found in 2013 by recreational spelunkers, a collection of bones was uncovered in a deep cave network in Johannesburg, South Africa. The cave system, known as Rising Star, had been well documented by other cavers; however, it appears few people had ever gone as far into the cave as these spelunkers had. Lee Berger, paleoanthropologist at University of Witwatersrand, in Johannesburg, immediately put out a call for what he termed “underground astronauts” to begin recovery and excavation of the fossil materials. Unlike other excavations, Berger and most other paleoanthropologists would not be able to access the site, as it was incredibly difficult to reach, and at some points there was only eight inches of space through which to navigate. The underground astronauts, all petite, slender female anthropologists, were the only ones who were able to access this remarkable site. Armed with small excavation tools and a video camera, which streamed the footage up to the surface, the team worked together and uncovered a total of 1,550 bones, representing at least 15 individuals, as seen in Figure 11.15. Later, an additional 131 bones, including an almost-complete cranium, were found in a nearby chamber of the cave, representing three more individuals (Figure 11.16). Berger called in a team of specialists to participate in what was dubbed “Paleoanthropology Summer Camp.” Each researcher specialized in a different portion of the hominin skeleton. With various specialists working simultaneously, more rapid analysis was possible of Homo naledi than most fossil discoveries.
The features of Homo naledi are well-documented due to the fairly large sample, which represents individuals of all sexes and a wide range of ages. The skull shape and features are very much like other members of the genus Homo, including features such as a sagittal keel and large brow, like Homo erectus, and a well-developed frontal lobe, similar to modern humans, yet the brain size is significantly smaller than its counterparts, at approximately 500 cc (560 cc for males and 465 cc for females). The teeth also exhibit features of later members of the genus Homo, such as Neanderthals, including a reduction in overall tooth size. Homo naledi also had unique shoulder anatomy and curved fingers, indicating similarities to tree-dwelling primates, which is very different from any other hominin yet found. The date on the species, which was obtained some time after the fossils were first analyzed, indicates that Homo naledi lived between 335,000 and 236,000 years ago. This has been perhaps the greatest shock of all about Homo naledi, as this places this fossil as a contemporary to modern Homo sapiens, despite the very primitive features it retains.

Other remarkable aspects of the find is the placement of the Homo naledi fossils and what it may suggest. To access the site, approximately 80 m from any known cave entrance or opening, a treacherous route would have had to have been taken—including moving through a portion that is just 25 cm wide at some points, known as “Superman’s Crawl.” The only way to get through this section is by crawling on your stomach with one arm by your side and the other raised above your head. Past Superman’s Crawl, a jagged wall known as the Dragon’s Back would have been very difficult to traverse. Below that, a narrow vertical chute would have eventually led down to the area where the fossils were discovered. While geology changes over time and the cave system likely has undergone its fair share, it is not likely that these difficult features arose after Homo naledi lived. This has made scientists curious as to how the bones ended up in the bottom of the cave system in the first place. It has been suggested that Homo naledi deposited the bones there, one way or another. Another competing idea is that a few individuals may have entered the cave system to escape a predator and then got stuck. To account for the sheer number of fossils, this would have had to happen multiple times. In contrast, if Homo naledi did deposit the bones, either through random disposal or intentional burial, this calls into question their symbolic behavior and other cultural traits, including the use of fire, to access a very dark cave system.
Homo floresiensis


In a small cave called Liang Bua, on the island of Flores, in Indonesia, a small collection of fossils were discovered beginning in 2003 (Figure 11.17). The fossil fragments represent as many as nine individuals, including a nearly complete female skeleton. The features of the skull are very similar to that of Homo erectus, including the presence of a sagittal keel, an arching brow ridges and nuchal torus, and the lack of a chin (Figure 11.18). Homo floresiensis, as the new species is called, had a brain size that was remarkably small at 400 cc, and recent genetic studies suggest a common ancestor with modern humans that predates Homo erectus.

The complete female skeleton, who was an adult, was approximately a meter tall and would have weighed just under 30 kg, which is significantly shorter and just a few kilograms more than the average, modern young elementary-aged child. A reconstructed comparison between an anatomically modern human and Homo floresiensis can be seen in Figure 11.19. The small size of the fossil has earned the species the nickname “the Hobbit.” Many questions have been asked about the stature of this species, as all of the specimens found also show evidence of diminutive stature and small brain size. Some explanations include pathology; however, this seems increasingly unlikely as all fossils found thus far demonstrate the same pattern. Another possible explanation lies in a biological phenomena seen in other animal species also found on the island and which date to a similar time period. This phenomenon, called , is due to limited food resources on an island, which can create a selective pressure for large-bodied species to be selected for smaller size, as an island would not have been able to support their larger-bodied cousins for a long period of time. This phenomenon is the cause of other unique species known to have lived on the island at the same time, including the miniature stegadon, a dwarf elephant species.
Homo floresiensis fossils have been dated to have lived on the island between 100,000 and at least 60,000 years ago. There is ongoing research and debate regarding Homo floresiensis’ dates of existence, with some researchers concluding that they lived on Flores until perhaps as recently as 17,000 years ago. Stone tools were also uncovered that have dates overlapping with those of the site and are similar in nature to other hominin stone tools found on the island of Flores. Homo floresiensis would have hunted a wide range of animals, including the miniature stegadon, giant rats, and other large rodents. Other animals on the island that could have threatened them include the giant komodo dragon. An interesting note about this island chain is that ancestors of Homo floresiensis would have had to access the open ocean in order to get there, as the nearest island is almost 10 km away, and there is little evidence to support that a land bridge connecting mainland Asia or Australia to the island would have been present. This would also have limited the number of other animals, including predators as well as human species, that would have had access to the island. Anatomically modern Homo sapiens arrived on the island around 30,000 years ago and may have lived there at the same time as Homo floresiensis, if some researchers’ later dates for Homo floresiensis’ occupation are correct. The modern population living on the island of Flores today believes that their ancestors came from the Liang Bua cave; however, recent genetic studies have determined they are not related to the Homo floresiensis species.
Homo naledi and Homo floresiensis are clear outliers when compared to their contemporary hominin species. Each has surprised paleoanthropologists for both their archaic traits in relatively modern times and their unique combination of traits seen in archaic species and modern species of humans. While these finds have been exciting, they have also completely upended the assumed trajectory of the human lineage, causing scientists to re-examine their previously held assumptions about hominin evolution and what it means to be modern. Add this to the developments being made using ancient DNA, other new fossil discoveries, and other innovations in paleoanthropology, and you see that our understanding of archaic Homo sapiens and others living during this time period is rapidly developing and changing. This is a true testament to the nature of science and the scientific method!
FOSSIL SPECIES SUMMARIES
| Hominin | archaic Homo sapiens |
| Dates | 600,000–200,000 years ago (although some regional variation) |
| Region(s) | Africa, Europe, and Asia |
| Famous discoveries | Broken Hill (Zambia), Atapuerca (Spain) |
| Brain size | 1,200 cc average |
| Dentition | Slightly smaller teeth in back of mouth, larger front teeth |
| Cranial features | Emerging forehead, wide nasal aperture, midfacial prognathism, no chin, projecting occipital region |
| Postcranial features | Robust skeleton |
| Culture | Varied regionally, but some continue to use Acheulean handaxe, others adopt Mousterian tool culture |
| Other | Lots of regional variation in this species |
| Hominin | Neanderthals |
| Dates | 150,000–35,000 years ago |
| Region(s) | Western Europe, Middle East, and Western Asia only |
| Famous discoveries | Shanindar (Iraq), La Chapelle-Aux-Saints (France) |
| Brian size | 1500 cc average |
| Dentition | Retromolar gap |
| Cranial features | Large brow ridge, midfacial prognathism, large infraorbital foramina, occipital bun |
| Postcranial features | Robust skeleton with short and stocky body, increased musculature, barrel chest |
| Culture | Mousterian tools often constructed using the Levallois technique |
| Hominin | Denisovans |
| Dates | 100,000–30,000 years ago |
| Region(s) | Siberia |
| Famous discoveries | Child’s finger bone and adult molar |
| Brain size | unknown |
| Dentition | unknown |
| Cranial features | unknown |
| Postcranial features | unknown |
| Culture | unknown |
| Other | Closely related to Neanderthals (genetically) |
| Species | Homo naledi |
| Dates | 335,000-235,000 years ago |
| Region(s) | South Africa |
| Famous Discoveries | Rising Star Cave |
| Brain size | 500 cc average |
| Dentition | Reduced tooth size |
| Cranial features | Sagittal keel, large brow, well-developed frontal region |
| Postcranial features | unknown |
| Culture | unknown |
| Species | Homo floresiensis |
| Dates | 100,000–60,000 years ago, perhaps as recently as 17,000 years ago |
| Region(s) | Lingua Bua, island of Flores, Indonesia |
| Famous discoveries | “The Hobbit” |
| Brian size | 400 cc average |
| Dentition | unknown |
| Cranial features | Sagittal keel, arching brow ridges, nuchal torus, no chin |
| Postcranial features | Very short stature (approximately 3.5 ft.) |
| Culture | Similar to other tools found on the island of Flores |
Review Questions
- What physical and cultural features are unique to archaic Homo sapiens? How are archaic Homo sapiens different in both physical and cultural characteristics from Homo erectus?
- Describe the specific changes to the brain and skull first seen in archaic Homo sapiens. Why does the shape of the skull change so dramatically from Homo erectus?
- What role did the shifting environment play in the adaptation of archaic Homo sapiens, including Neanderthals? Discuss at least one physical feature and one cultural feature that would have assisted these groups in surviving the changing environment.
- In your opinion, which of the hypotheses concerning the disappearance of the Neanderthals best incorporates the available evidence? Why?
- What does the regional variation in archaic Homo sapiens represent in terms of the broader story of our species’ evolution?
- Describe the issues raised by the discoveries of Homo naledi and Homo floresiensis in the understanding of the story of the evolution of Homo sapiens.
Key Terms
5 prime end: A nucleic acid strand that terminates at the chemical group attached to the fifth carbon in the sugar-ring.
3 prime end: A nucleic acid strand that terminates at the hydroxyl (-OH) chemical group attached to the third carbon in the sugar-ring.
Allele: Each of two or more alternative forms of a gene that arise by mutation and are found at the same place on a chromosome.
Anthropocentrism: A way of thinking that assumes humans are the most important species and leads to interpreting the world always through a human lens. Species-centric science and thought.
Coalescent methods: These are models which allow for inference of how genetic variants sampled from a population may have originated from a common ancestor
Cortex: The outside, or rough outer covering, of a rock. Usually the cortex is removed during the process of stone tool creation.
Deamination: The chemical process that results in the conversion of Cytosine to uracil, which results in Cytosine to Thymine conversions during sequencing.
Divergence time: A measure of how long two genomic sequences have been changing independently.
Endogenous aDNA: A form of ancient DNA in which DNA originates from the specimen being examined.
Ethnocentric: Applying negative judgments to other cultures based on comparison to one’s own.
Exogenous DNA: DNA that originates from sources outside of the specimen you are trying to sequence.
Flexed position: Fetal position, in which the legs are drawn up to the middle of the body and the arms are drawn toward the body center. Intentional burials are often found in the flexed body position.
Foraminifera: Microscopic single-celled organisms with a shell that are common in all marine environments. The fossil record of foraminifera extends back well over 500 million years.
Glaciation: A glacial period, or time when a large portion of the world is covered by glaciers and ice sheets.
Globular: Round-shaped, like a globe.
Grave goods: Items included with a body at burial. Items may signify occupation or hobbies, social status, or level of importance in the community, or they may be items believed necessary for the afterlife.
Haft: A handle. Also used as a verb—to attach a handle to an item, such as a stone tool.
Haplotype: A set of genetic variants located on a single stretch of the genome. This unique combination of variants on a stretch of the genome can be used to differentiate groups that will have different combinations of variants.
Heterozygosity: A measure of how many genes within a diploid genome are made up of more than one variant for a gene.
High-coverage sequences: These are genomic sequences which have been sequenced multiple times to ensure that the sequence produced is a true reflection of the genomic sequence, and reduce the likelihood that the sequence has sequencing errors as a result of the the sequencing process.
Homozygosity: A measure of how many genes within a diploid genome are made up of more than the same variant for a gene.
Ice core: A colundular sample of ice that is removed from an ice sheet. The annual buildup of snow and ice can be studied and interpreted to better understand climate change, as well as local environmental shifts.
Infraorbital foramina: Small holes on the maxilla bone of the face that allows nerves and blood to reach the skin.
Insular dwarfing: A form of dwarfism that occurs when a limited geographic region, such as an island, causes a large-bodied animal to be selected for a smaller body size.
Interglacial: A warmer period between two glacial time periods.
Introgressed genes: This is the movement of genes from one species to the gene pool of another species through hybridization between the species and backcross into the parental population by hybrid offspring.
Levallois technique: A distinctive technique of stone tool manufacturing used by archaic Homo sapiens, including Neanderthals. The technique involves the preparation of a core and striking edges off in a regular fashion around the core. Then a series of similarly sized pieces can be removed, which can then be turned into different tools.
Midfacial prognathism: A forward projection of the nose, or middle facial region. Usually associated with Neanderthals.
Mousterian tools: The stone tool industry of Neanderthals and their contemporaries in Africa and Western Asia. Mousterian tools are known for a diverse set of flake tools, which is different than the large bifacial tools of the Acheulean industry.
Nasal aperture: The opening for the nose visible on a skull. Often pear- or heart-shaped.
Non-synonymous mutations: These are changes that also occur in the protein-coding region of the genome but don’t result in a change in amino acid sequence of the protein being produced.
Occipital bun: A prominent bulge or projection on the back of the skull, specifically the occipital bone. This is a feature present only on Neanderthal skulls.
Ochre: A natural clay pigment mixed with ferric oxide and clay and sand. Ranges in color from brown to red to orange.
Recombination: This is the process of exchange of DNA between two strands to produce new sequence arrangements.
Retracted face: A face that is flatter.
Retromolar gap: A space behind the last molar and the end of the jaw. This is a feature present only on Neanderthals. It also occurs through cultural modification in modern humans who have had their third molars, or wisdom teeth, removed.
Sediment core: A colundular sample of soil and sediments that can be analyzed to study plant and animal presence in a location, or more broadly to determine a regional environment.
Synonymous mutations: Mutations that occur in the protein-coding region of the genome and result in a change in the amino acid sequence of the protein produced.
About the Authors
Amanda Wolcott Paskey
Amanda Wolcott Paskey is an anthropology professor at Cosumnes River College in Sacramento, California. She earned her B.A. and M.A. in anthropology from the University of California, Davis. Her speciality in anthropology is archaeology; however, she was trained in a holistic program and most of her teaching load is in biological anthropology. She is currently working on analyzing a post–gold rush era archaeological site, in Sacramento, with colleagues and students. This project has given her many opportunities to engage in sharing archaeology with a public audience, including local school children and Sacramentans interested in local history.
AnneMarie Beasley Cisneros

AnneMarie Beasley Cisneros is an anthropology professor at American River College in Sacramento, California. Trained as a four-field anthropologist, she earned her B.A. and M.A. in anthropology from California State University, Sacramento. She regularly teaches biological anthropology, among other courses, and is currently engaged in applied anthropology work in community development with historically underserved communities. She most recently has particularly enjoyed facilitating her students’ involvement in projects serving Sacramento’s Latino and immigrant Mexican populations.
About the Special Topic: Ancient DNA Author

Robyn Humphreys is a biological anthropologist based in the archaeology department at the University of Cape Town. Her MSc focused on the role of hybridization in human evolution. She is now pursuing her Ph.D., which will involve looking at the relationship between archaeologists and communities in relation to research on human remains from historical sites in Cape Town.
For Further Exploration
Anne and Bernard Spitzer Hall of Human Origins—American Museum of Natural History https://www.amnh.org/exhibitions/permanent-exhibitions/anne-and-bernard-spitzer-hall-of-human-origins
“Dawn of Humanity,” PBS documentary, 2015
“DNA Clues to Our Inner Neanderthal,” TED Talk by Svante Pääbo, 2011. https://www.ted.com/talks/svante_paeaebo_dna_clues_to_our_inner_neanderthal?language=en
“The Dirt” Podcast, Episode 30 “The Human Family Tree (Shrub? Crabgrass? Tumbleweed?), Part 3: Very Humany Indeed”
https://thedirtpod.com/episodes//episode-30-the-human-family-tree-shrub-crabgrass-tumbleweed-part-3
E Fossil games and activities http://www.efossils.org/page/games-and-activities
“Hobbits on Flores, Indonesia” Smithsonian Human Origins http://humanorigins.si.edu/research/asian-research-projects/hobbits-flores-indonesia
Shanindar 3—Neanderthal Skeleton—Smithsonian Human Origins http://humanorigins.si.edu/evidence/human-fossils/shanidar-3-neanderthal-skeleton
Smithsonian’s Human Origins Program Facebook page (@smithsonian.humanorigins) https://www.facebook.com/smithsonian.humanorigins/
Paleoartist Brings Human Evolution to Life—Elisabeth Daynés https://www.smithsonianmag.com/science-nature/bringing-human-evolution-life-180951155/
Green, R. E., J. Krause, A. W. Briggs, T. Maricic, U. Stenzel, M. Kircher, et al. 2010. “A Draft Sequence of the Neanderthal Genome.” Science 328 (5979): 710–722.
Gibbons, A. 2014. “Neanderthals and Moderns Made Imperfect Mates.” Science 343 (6170): 471–472. doi: 10.1126/science.343.6170.471.
Huerta-Sánchez, E., X. Jin, Z. Bianba, B. M. Peter, N. Vinckenbosch, Y. Liang, et al. 2014. Altitude Adaptation in Tibetans Caused by Introgression of Denisovan-like DNA. Nature 512 (7513): 194.
Jeong, C., G. Alkorta-Aranburu, B. Basnyat, M. Neupane, D. B. Witonsky, J. K. Pritchard, C. M. Beatl, and A. Di Rienzo. 2014. “Admixture Facilitates Genetic Adaptations to High Altitude in Tibet.” Nature Communications 5: 3281. doi: 10.1038/ncomms4281
Krings, M., A. Stone, R. W. Schmitz, H. Krainitzki, M. Stoneking, and S. Pääbo. 1997. “Neanderthal DNA Sequences and the Origin of Modern Humans.” Cell 90 (1): 19–30.
Krouse, J., Q. Fu, J. M. Good, B. Viola, M. V. Shunkov, A. P. Derevianko, and S. Pääbo. 2010. “The Complete Mitochondrial DNA Genome of an Unknown Hominin from Southern Siberia.” Nature 464: 894–897.
Prüfer, K., F. Racimo, N. Patterson, F. Jay, S. Sankararaman, S. Sawyer, et al. 2014. “The Complete Genome Sequence of a Neanderthal from the Altai Mountains.” Nature 505 (7481): 43-49.
Racimo, F., S. Sankararaman, R. Nielsen, and E. Huerta-Sánchez. 2015. Evidence for Archaic Adaptive Introgression in Humans. Nature Reviews Genetics 16 (6): 359-371.
Sankararaman, Sriram, et al. “The genomic landscape of Neanderthal ancestry in present-day humans.” Nature 507.7492 (2014): 354.
Vernot, B., S. Tucci, J. Kelso, J. G. Schraiber, A. B. Wolf, R. M. Gittelman, et al. 2016. “Excavating Neanderthal and Denisovan DNA from the Genomes of Melanesian Individuals.” Science 352 (6282): 235–239.
References
Atkinson, E. G., A. J. Audesse, J. A. Palacios, D. M. Bobo, A. E. Webb, S. Ramachandran, B. M. Henn. 2018. “No Evidence for Recent Selection at FOXP2 among Diverse Human Populations.” Cell 174 (6): 1424–1435.
Berger, T. D, and E. Trinkaus. 1995. “Patterns of Trauma among the Neanderthals.” Journal of Archaeological Science 22 (6): 841–852.
Bocherens, Hervé, Daniel Billiou, André Mariotti, Marylène Patou-Mathis, Marcel Otte, Dominique Bonjean, and Michel Toussaint. 1999. “Palaeoenvionmental and Palaeodietary Implications of Isotopic Biogeochemistry of Last Interglacial Neanderthal and Mammal Bones in Scladina Cave (Belgium).” Journal of Archaeological Science 26 (6): 599–607.
Bocquet-Appel, J. P., and A. Degioanni. 2013. “Neanderthal Demographic Estimates.” Current Anthropology 54 (S8): S202–S213.
Churchill, S. E. 1998. “Cold Adaptations, Heterochrony, and Neanderthals.” Evolutionary Anthropology 7: 46–61.
Churchill, S. E., R. G. Franciscus, H. A. McKean-Peraza, J. A. Daniel, and B. R. Warren. 2009. “Shanidar 3 Neanderthal Rib Puncture Wound and Paleolithic Weaponry.” Journal of Human Evolution 57 (2): 163–178.
Degioanni A., C. Bonenfant, S. Cabut, and S. Condemi. 2019. Living on the Edge: Was Demographic Weakness the Cause of Neanderthal Demise? PLoS ONE 14 (5): e0216742.
doi: 10.1371/journal.pone.0216742
Dirks, P. H. G. M., E. M. Roberts, H. Hilbert-Wolf, J. D. Kramers, J. Hawks, A. Dosseto, M. Duval, et al. 2017. “The Age of Homo naledi and Associated Sediments in the Rising Star Cave, South Africa.” eLife 6 (24231). doi: 10.7554/eLife.24231
el-Showk, S. 2019. “Neanderthal Clues to Brain Evolution in Humans.” Nature 571: S10–S11. doi: 10.1038/d41586-019-02210-6
Enard, D. and D. A. Petrov. 2018. “Evidence that RNA Viruses Drove Adaptive Introgression between Neanderthals and Modern Humans.” Cell 175 (2): 360–371. doi: 10.1016/j.cell.2018.08.034
Frayer, D. W., M. Lozano, J. M. Bermudez de Castro, E. Carbonell, J.-L. Arsuaga, J. Radovcic, I. Fiore, and L. Bondioli. 2012. “More Than 500,000 Years of Righthandness in Europeans.” Laterality 17 (1): 51–69.
Gilpin, W., M. W. Feldman, and K. Aoki. 2016. “An Ecocultural Model Predicts Neanderthal Extinction through Competition With Modern Humans.” Proceedings of the National Academy of Sciences 113 (8): 2134–2139.
Golovanova, L.V., V. B. Doronichev, N. E. Cleghorn, M. A. Koulkova, T. V. Sapelko, and M. S. Shackley. 2010. “Significance of Ecological Factors in the Middle to Upper Paleolithic Transition.” Current Anthropology 51 (5): 655-691.
Houldcroft, C. J., and S. J. Underdown. 2016. “Neanderthal Genomics Suggests a Pleistocene Time Frame for the First Epidemiologic Transition.” American Journal of Physical Anthropology 160 (3): 379–388.
Jaouen, K., A. Le Cabec, F. Welker, J. Hublin, M. Soressi, and S. Talamo. 2019. “Exceptionally High δ15N Values in Collagen Single Amino Acids Confirm Neanderthals as High-Trophic Level Carnivores.” Proceedings of the National Academy of Sciences 116 (11): 4928–4933. doi: 10.1073/pnas.1814087116
Kortlandt, Adriaan. 2002. “Neanderthal Anatomy and the Use of Spears.” Evolutionary Anthropology 11 (5): 183–184.
Macdonald, K., W. Roebroeks, and A. Verpoorte. 2009. “An Energetics Perspective on the Neanderthal Record.” In The Evolution of Hominin Diets: Integrating Approaches to the Study of Palaeolithic Subsistence, edited by J.-J. Hublin and M. P. Richards, pp. 211–e220. Dordrecht, Germany: Springer.
Mendez, F. L., G. D. Poznik, S. Castellano, and C. D. Bustamante. 2016. “The Divergence of Neanderthal and Modern Human Y Chromosomes.” American Journal of Human Genetics 98 (4): 728–734.
Pearce, E., C. M. Stringer, and R. I. M. Dunbar. 2013. “New Insights into Differences in Brain Organizations between Neanderthals and Anatomically Modern Humans.” Proceedings of the Royal Society B: Biological Sciences 280 (1758): 20130168. doi: 10.1098/rspb.2013.0168
Staubwasser, M., V. Drăguʂin, B. P. Onac, S. Assonov, V. Ersek, D. O. Hoffman, and D. Veres. 2018. “Impact of Climate Change on the Transition of Neanderthals to Modern Humans in Europe.” Proceedings of the National Academy of Sciences 115 (37): 9116–9121.
doi: 10.1073/pnas.1808647115
Stringer, C. B. 1996. “Current Issues in Modern Human Origins.” In Contemporary Issues in Human Evolution, edited by W. E. Meickle, F. C. Howell, and N. G. Jablonski, pp. 115–134. San Francisco: California Academy of Sciences.
Trinkaus, E. 2006. “Modern Human versus Neandertal Evolutionary Distinctiveness,” Current Anthropology 47, no. 4 (August 2006): 597-620. https://doi.org/10.1086/504165
Trinkaus, E. 2007. “European Early Modern Humans and the Fate of the Neanderthals.” PNAS May 1, 2007 104 (18) 7367-7372; https://doi.org/10.1073/pnas.0702214104
Venner, S. J. 2018. “A New Estimate for Neanderthal Energy Expenditure.” CUNY Academic Works. https://academicworks.cuny.edu/hc_sas_etds/327
Weaver, T. D., and J. Hublin. 2009. “Neanderthal Birth Canal Shape and the Evolution of Human Childbirth.” Proceedings of the National Academy of Sciences 106 (20): 8151–8156.
Wiẞing, C. H. Rougier, I. Crevecoer, M. Germonpré, Y. Naito, P. Semal, and H. Bocherens. 2015. “Isotopic Evidence for Dietary Ecology of Late Neanderthals in Northwestern Europe.” Quaternary International 411 (A): 327–345.
doi: 10.1016/j.quaint.2015.09.091
Wong, K. 2015. “Different yet Alike.” Scientific American 312, 2, 6 (February 2015) doi:10.1038/scientificamerican0215-6
Acknowledgments
The authors would like to extend their thanks to Cassandra Gilmore and Anna Goldfield for thoughtful and insightful suggestions on this chapter.
Figure Attributions
Figure 11.1 Big head primitive caveman nose man bone cave at Max Pixel has been designated to the public domain (CC0).
Figure 11.2 All paleotemps by Glen Fergus is used under a CC BY-SA 3.0 License.
Figure 11.3 Homo erectus, archaic Homo sapiens, and anatomically modern Homo sapiens table original to Explorations: An Open Invitation to Biological Anthropology is under a CC BY-NC 4.0 License.
Figure 11.4 Homo heidelbergensis Cranium Broken Hill 1 (Rhodesian Man) by ©BoneClones is used by permission and available here under a CC BY-NC 4.0 License.
Figure 11.5 Homo heidelbergensis Skull Atapuerca 5 by ©BoneClones is used by permission and available here under a CC BY-NC 4.0 License.
Figure 11.6 Neanderthal distinguishing features table original to Explorations: An Open Invitation to Biological Anthropology is under a CC BY-NC 4.0 License.
Figure 11.7 Neanderthal Skeleton Articulated by ©BoneClones is used by permission and available here under a CC BY-NC 4.0 License.
Figure 11.8 Nucléus Levallois La-Parrilla by José-Manuel Benito Álvarez is used under a CC BY-SA 2.5 License,
Figure 11.9 NHM – Levalloiskern by Wolfgang Sauber is used under a CC BY-SA 4.0 License.
Figure 11.10a Homo neanderthalensis Shanidar 1 Skull by ©BoneClones is used by permission and available here under a CC BY-NC 4.0 License.
Figure 11.10b Shanidar 1 by Smithsonian [exhibit: Human Evolution Evidence, Human Fossils, Species, Homo neanderthalensis] is copyrighted and used for educational and non-commercial purposes as outlined by the Smithsonian.
Figure 11.11 Neandertala homo, modelo en Neand-muzeo (image was taken in the Neanderthal Museum, Mettmann, Germany) by UNiesert is used under a CC BY-SA 3.0 License.
Figure 11.12 DNA extraction by Robyn Humphreys original to Explorations: An Open Invitation to Biological Anthropology is under a CC BY-NC 4.0 License.
Figure 11.13 Homo sapiens lineage by Dbachmann is used under a CC BY-SA 4.0 License.
Figure 11.14 Introgressed Neanderthal DNA in a modern human genome by Robyn Humphreys original to Explorations: An Open Invitation to Biological Anthropology is under a CC BY-NC 4.0 License.
Figure 11.15 Dinaledi skeletal specimens (Figure 1) by Berger, Lee R. et al. 2015. “Homo naledi, a new species of the genus Homo from the Dinaledi Chamber, South Africa.” eLife 2015;4:e09560 (DOI: 10.7554/eLife.09560) is used under a CC BY 4.0 License.
Figure 11.16 LES1 Cranium (Figure 5) by Hawks et al. 2017. “New fossil remains of Homo naledi from the Lesedi Chamber, South Africa”. eLife 2017;6:e24232. (DOI:10.7554/eLife.24232) is used under a CC BY 4.0 License.
Figure 11.17 Homo floresiensis cave by Rosino is used under a CC BY-SA 2.0 License.
Figure 11.18 Homo floresiensis Skull (Flores Skull LB1) by ©BoneClones is used by permission and available here under a CC BY-NC 4.0 License.
Figure 11.19 Anatomically modern human and Homo floresiensis reconstruction original to Explorations: An Open Invitation to Biological Anthropology by Mary Nelson is under a CC BY-NC 4.0 License.
Applying negative judgments to other cultures based on comparison to one’s own.
A way of thinking that assumes humans are the most important species and leads to interpreting the world always through a human lens. Species-centric science and thought.
A glacial period, or time when a large portion of the world is covered by glaciers and ice sheets.
A warmer period between two glacial time periods.
Single-celled marine organisms with shells.
Core samples taken from lake beds or other water sources for analysis of their pollen.
A colundular sample of ice that is removed from an ice sheet. The annual buildup of snow and ice can be studied and interpreted to better understand climate change, as well as local environmental shifts.
Round-shaped, like a globe.
A face that is flatter.
The opening for the nose visible on a skull. Often pear- or heart-shaped.
A forward projection of the nose, or middle facial region. Usually associated with Neanderthals.
A space behind the last molar and the end of the jaw. This is a feature present only on Neanderthals. It also occurs through cultural modification in modern humans who have had their third molars, or wisdom teeth, removed.
A prominent bulge or projection on the back of the skull, specifically the occipital bone. This is a feature present only on Neanderthal skulls.
Small holes on the maxilla bone of the face that allows nerves and blood to reach the skin.
The stone tool industry of Neanderthals and their contemporaries in Africa and Western Asia. Mousterian tools are known for a diverse set of flake tools, which is different than the large bifacial tools of the Acheulean industry.
A distinctive technique of stone tool manufacturing used by Archaic Homo sapiens, including Neanderthals. The technique involves the preparation of a core and striking edges off in a regular fashion around the core. Then a series of similarly sized pieces can be removed, which can then be turned into different tools.
The outside, or rough outer covering, of a rock. Usually the cortex is removed during the process of stone tool creation.
A handle. Also used as a verb—to attach a handle to an item, such as a stone tool.
Fetal position, in which the legs are drawn up to the middle of the body and the arms are drawn toward the body center. Intentional burials are often found in the flexed body position.
Items included with a body at burial. Items may signify occupation or hobbies, social status, or level of importance in the community, or they may be items believed necessary for the afterlife.
A natural clay pigment mixed with ferric oxide and clay and sand. Ranges in color from brown to red to orange.
These are genomic sequences which have been sequenced multiple times to ensure that the sequence produced is a true reflection of the genomic sequence, and reduce the likelihood that the sequence has sequencing errors as a result of the the sequencing process.
A form of ancient DNA in which DNA originates from the specimen being examined.
DNA that originates from sources outside of the specimen you are trying to sequence.
The chemical process that results in the conversion of Cytosine to uracil, which results in Cytosine to Thymine conversions during sequencing.
A nucleic acid strand that terminates at the chemical group attached to the fifth carbon in the sugar-ring.
A nucleic acid strand that terminates at the hydroxyl (-OH) chemical group attached to the third carbon in the sugar-ring.
A set of genetic variants located on a single stretch of the genome. This unique combination of variants on a stretch of the genome can be used to differentiate groups that will have different combinations of variants.
These are models which allow for inference of how genetic variants sampled from a population may have originated from a common ancestor
A change in the DNA sequence that codes for amino acids in a protein sequence, but does not change the encoded amino acid.
A point mutation that causes a change in the resulting protein.
A measure of how many genes within a diploid genome are made up of more than one variant for a gene.
A non-identical DNA sequence found in the same gene location on a homologous chromosome, or gene copy, that codes for the same trait but produces a different phenotype. In other words, a gene variant.
A measure of how many genes within a diploid genome are made up of more than the same variant for a gene.
A measure of how long two genomic sequences have been changing independently.
This is the process of exchange of DNA between two strands to produce new sequence arrangements.
This is the movement of genes from one species to the gene pool of another species through hybridization between the species and backcross into the parental population by hybrid offspring.
A form of dwarfism that occurs when a limited geographic region, such as an island, causes a large-bodied animal to be selected for a smaller body size.
